HMGB1-TLR4-RAGE Signaling in Sterile Inflammation: Mechanisms, Methods, and Therapeutic Targeting
This comprehensive review elucidates the pivotal role of HMGB1 signaling through Toll-like Receptor 4 (TLR4) and Receptor for Advanced Glycation End-products (RAGE) in driving sterile inflammation.
HMGB1-TLR4-RAGE Signaling in Sterile Inflammation: Mechanisms, Methods, and Therapeutic Targeting
Abstract
This comprehensive review elucidates the pivotal role of HMGB1 signaling through Toll-like Receptor 4 (TLR4) and Receptor for Advanced Glycation End-products (RAGE) in driving sterile inflammation. Targeting researchers, scientists, and drug development professionals, it systematically explores foundational biology and molecular pathways (Intent 1), details state-of-the-art experimental methodologies for pathway interrogation (Intent 2), addresses common technical challenges and optimization strategies (Intent 3), and critically compares current therapeutic approaches and validation models (Intent 4). The article synthesizes the latest research to provide a roadmap for developing targeted interventions in sterile inflammatory diseases such as ischemia-reperfusion injury, neurodegeneration, and autoimmune disorders.
Decoding HMGB1-TLR4-RAGE Axis: Core Mechanisms in Sterile Inflammatory Signaling
High Mobility Group Box 1 (HMGB1) is a prototypical damage-associated molecular pattern (DAMP) or alarmin, playing a pivotal role in initiating and perpetuating sterile inflammation. Its biological activity is critically determined by its redox state, which dictates receptor binding preference—primarily between Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End products (RAGE). This whitepaper details the structural basis, release mechanisms, and redox chemistry of HMGB1, providing a technical foundation for research within the broader thesis of HMGB1-TLR4-RAGE signaling in sterile inflammatory diseases.
Structure of HMGB1
HMGB1 is a 215-amino acid, non-histone chromosomal protein organized into three distinct domains:
- A-Box (aa 1-85): Contains a redox-sensitive cysteine at position 23 (C23). Acts as a competitive antagonist to full-length HMGB1.
- B-Box (aa 88-162): The primary pro-inflammatory domain, containing a redox-sensitive cysteine at position 45 (C45).
- Acidic Tail (C-terminal, aa 186-215): Rich in aspartic and glutamic acids, modulating DNA binding and inflammation.
The two critical cysteines, C23 and C45, undergo post-translational redox modifications that define HMGB1's extracellular signaling functions.
Release Mechanisms
HMGB1 can be released from cells via both passive and active pathways, summarized in Table 1.
Table 1: HMGB1 Release Mechanisms
| Mechanism | Cell Type/Trigger | Key Features | Kinetics |
|---|---|---|---|
| Passive Release | Necrotic cells | Uncontrolled leakage due to loss of membrane integrity. Retains pro-inflammatory potential. | Immediate |
| Active Secretion | Activated immune cells (macrophages, monocytes, dendritic cells) | Regulated process involving translocation to cytosol and exocytosis. Requires acetylation/nucleus-to-cytoplasm shuttling. | 12-24 hrs post-stimulus |
| Pyroptotic Release | Cells undergoing pyroptosis | Gasdermin D pore formation allows passive leakage during programmed necrosis. | Post-inflammasome activation |
Experimental Protocol: In Vitro HMGB1 Release from LPS-Stimulated Macrophages
- Cell Culture: Seed RAW 264.7 or primary bone marrow-derived macrophages (BMDMs) in 12-well plates (2.5 x 10^5 cells/well).
- Stimulation: Treat cells with ultrapure LPS (100 ng/mL) for 16-24 hours.
- Inhibition (Optional): Pre-treat with acetyltransferase inhibitor (e.g., anacardic acid, 50 µM) or export inhibitor (ethyl pyruvate, 10 mM) for 1 hour prior to LPS.
- Sample Collection: Centrifuge culture supernatant at 500 x g for 5 min to remove debris. Collect cell lysate using RIPA buffer.
- Detection: Analyze HMGB1 levels in supernatant and lysate via Western Blot (primary anti-HMGB1 antibody, 1:2000 dilution) and quantify via ELISA.
Redox States and Signaling Specificity
The redox state of C23 and C45 creates distinct HMGB1 isoforms with unique receptor affinities (Table 2).
Table 2: HMGB1 Redox Isoforms and Functions
| Isoform Name | C23 State | C45 State | C106 State | Primary Receptor | Biological Function |
|---|---|---|---|---|---|
| Fully Reduced (fr-HMGB1) | -SH | -SH | -SH | CXCR4 | Chemotactic activity, promotes tissue repair. |
| Disulfide HMGB1 (ds-HMGB1) | -SH | -S-S- (with C23) | -SH | TLR4 / MD-2 | Potent pro-inflammatory cytokine, induces TNF/IL-6 release. |
| Terminally Oxidized (ox-HMGB1) | -SOx | -SOx | -SOx | RAGE | No cytokine activity; may promote immune tolerance and autophagy. |
Experimental Protocol: Redox-State Determination via Alkylation and Non-Reducing SDS-PAGE
- Sample Alkylation: Treat cell supernatant or purified HMGB1 with iodoacetamide (IAM, 20 mM final concentration) for 30 min in the dark. IAM alkylates free thiols (-SH), preventing post-lysis oxidation.
- Protein Precipitation: Use TCA/acetone precipitation to concentrate and desalt samples.
- Electrophoresis: Resuspend protein pellets in non-reducing Laemmli buffer (without β-mercaptoethanol or DTT). Run samples on a 12% SDS-PAGE gel.
- Analysis: Western Blot with anti-HMGB1 antibody. Different redox states exhibit different electrophoretic mobilities: fr-HMGB1 migrates faster, ds-HMGB1 shows intermediate migration, ox-HMGB1 migrates slowest.
Signaling Pathways: Integration with TLR4 and RAGE
The redox-dependent engagement of HMGB1 with TLR4 or RAGE initiates distinct but overlapping signaling cascades.
Diagram 1: HMGB1 Redox-Dependent Receptor Signaling
Title: HMGB1 Redox States Dictate Receptor Choice and Signaling
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for HMGB1 Studies
| Reagent / Material | Function / Application | Example / Note |
|---|---|---|
| Recombinant HMGB1 Isoforms | Provide defined redox states for in vitro stimulation assays. | fr-HMGB1, ds-HMGB1, ox-HMGB1 (commercially available or prepared in-house). |
| Site-Specific HMGB1 Antibodies | Detect HMGB1 and differentiate localization or modifications. | Anti-HMGB1 (general), anti-acetyl-HMGB1, antibodies to specific redox forms. |
| Receptor Antagonists/Inhibitors | To dissect specific signaling pathways in vitro/vivo. | TAK-242 (TLR4 inhibitor), FPS-ZM1 (RAGE inhibitor), AMD3100 (CXCR4 antagonist). |
| Thiol Alkylating Agents | To "trap" and preserve the in vivo redox state of HMGB1 for analysis. | Iodoacetamide (IAM), N-ethylmaleimide (NEM). Must be used immediately on fresh samples. |
| Redox Buffering Systems | To generate and maintain specific HMGB1 redox isoforms in vitro. | Glutathione redox couples (GSH/GSSG), DTT, hydrogen peroxide (H₂O₂). |
| HMGB1 ELISA Kits | Quantify total HMGB1 release in biological fluids (serum, supernatant). | Does not distinguish redox states. |
| Gasdermin D Inhibitor | To specifically block pyroptotic HMGB1 release. | Necrosulfonamide or disulfiram. |
| HMGB1 A-Box Protein | Acts as a functional antagonist in control experiments. | Used to block HMGB1 activity competitively. |
Diagram 2: Workflow for HMGB1 Redox State Analysis
Title: Experimental Workflow for HMGB1 Redox-State Detection
HMGB1 is a master regulator of sterile inflammation whose pleiotropic functions are exquisitely controlled by its redox state. The disulfide isoform serves as the key TLR4 agonist driving cytokine storm, while the fully reduced and terminally oxidized forms engage CXCR4 and RAGE, respectively, modulating chemotaxis and resolution. Precise experimental delineation of its structure, active release, and redox speciation is fundamental to understanding its role in disease pathogenesis and for developing targeted therapies that block its pathological signaling while sparing its reparative functions.
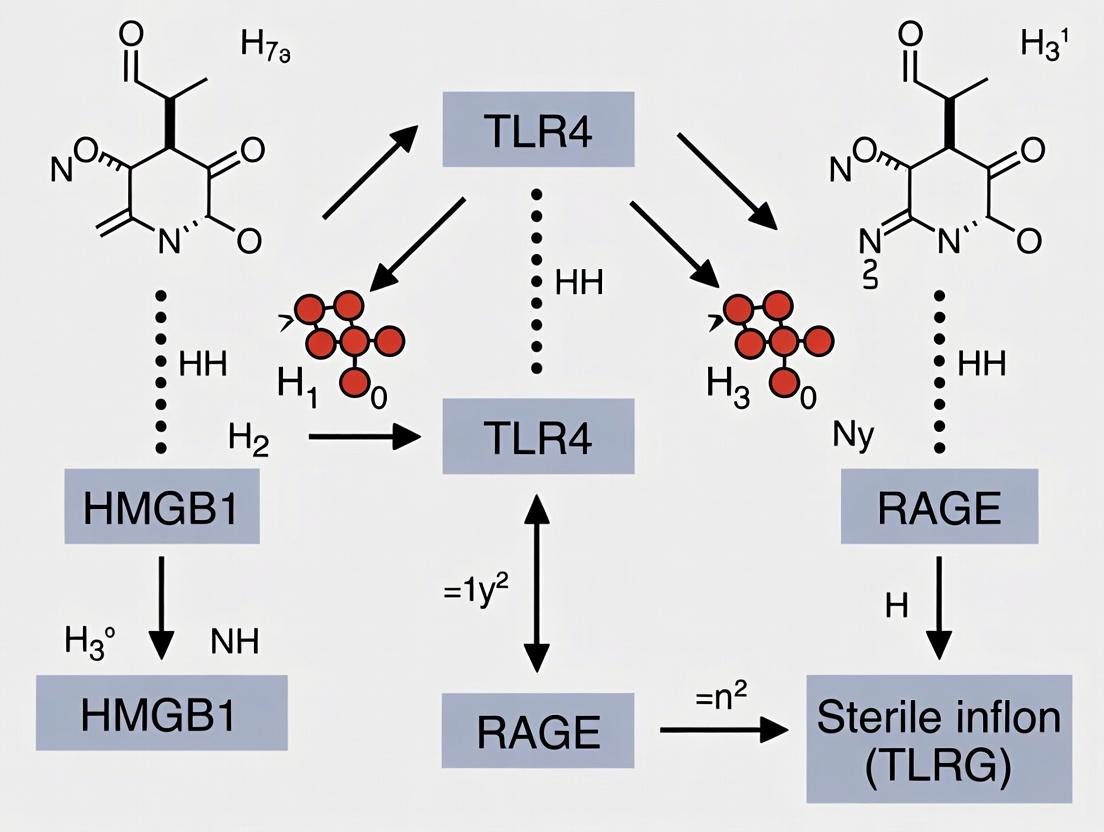
Within the framework of sterile inflammation research, the damage-associated molecular pattern (DAMP) HMGB1 is a master regulator, initiating potent inflammatory responses via two primary receptor systems: Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End-products (RAGE). Understanding the distinct and overlapping signaling complexes and downstream cascades triggered by these receptors is critical for developing targeted therapeutics. This whitepaper provides a technical delineation of TLR4 and RAGE signaling, focusing on HMGB1-mediated activation in sterile inflammatory conditions.
Receptor Complex Architecture and Ligand Binding
TLR4 Signaling Complex
TLR4 activation requires a multi-step assembly of core and accessory proteins. HMGB1, often in complex with other ligands like LPS or IL-1β, initiates signaling.
Core Complex Formation:
- MD-2 Binding: HMGB1 binding promotes the dimerization of the TLR4-MD-2 complex.
- TIR Domain Recruitment: Dimerized TLR4 cytoplasmic TIR domains recruit adaptor molecules: MyD88 (for the MyD88-dependent pathway) or TRIF (for the TRIF-dependent pathway), via the bridging adaptors TIRAP and TRAM, respectively.
RAGE Signaling Complex
RAGE is a multi-ligand transmembrane receptor of the immunoglobulin superfamily. Its signaling complex is more fluid and context-dependent.
Core Complex Formation:
- Ligand Binding & Oligomerization: HMGB1 binding induces RAGE oligomerization (dimerization or higher-order clustering).
- Cytoplasmic Tail Interactions: The short, highly charged cytoplasmic domain lacks intrinsic kinase activity but directly interacts with key signaling effectors like DIAPH1 (Diaphanous 1) and ERK1/2.
Table 1: Comparative Receptor Complex Characteristics
| Feature | TLR4 | RAGE |
|---|---|---|
| Receptor Family | Toll-like Receptor (TLR) | Immunoglobulin (Ig) Superfamily |
| Core Co-Receptor | MD-2 | Not Applicable (binds ligand directly) |
| Ligand Binding | Dimerization-dependent | Oligomerization-dependent |
| Key Adaptor Proteins | MyD88, TIRAP, TRIF, TRAM | DIAPH1 (direct binder) |
| Primary Docking Site | TIR Domain | Cytoplasmic Tail |
| Complex Stability | High, defined sequence | Dynamic, variable composition |
Downstream Signaling Cascades
TLR4 Downstream Pathways
TLR4 signaling bifurcates into two major pathways originating from different cellular compartments.
MyD88-Dependent Pathway (Plasma Membrane):
- Kinase Cascade: MyD88 recruits IRAK4, which phosphorylates IRAK1. IRAK1 interacts with TRAF6, leading to activation of TAK1.
- Key Transcription Factors: TAK1 activates the IKK complex (degrading IκB) to activate NF-κB, and the MAPK pathways (JNK, p38) to activate AP-1.
- Outcome: Rapid pro-inflammatory cytokine production (TNF-α, IL-6, IL-1β).
TRIF-Dependent Pathway (Endosome):
- Kinase Cascade: TRIF recruits TRAF3 and RIPK1, leading to TBK1/IKKε activation.
- Key Transcription Factor: TBK1/IKKε phosphorylates IRF3, promoting its dimerization and nuclear translocation.
- Outcome: Type I Interferon (IFN-β) and late-phase NF-κB activation.
RAGE Downstream Pathways
RAGE signaling is more linear and directly coupled to fundamental cellular processes.
Core Signaling Cascade:
- Ras-MAPK Activation: Oligomerized RAGE directly activates Ras (via unknown intermediaries), leading to sequential activation of Raf, MEK, and ERK1/2.
- DIAPH1-Dependent Actin Remodeling: The cytoplasmic tail binds DIAPH1, activating Rho GTPases (Rac1, Cdc42) to drive cytoskeletal reorganization.
- Key Transcription Factors: Sustained ERK1/2 and p38 MAPK activation leads to activation of NF-κB, AP-1, and CREB.
- Outcome: Pro-inflammatory gene expression, increased cellular oxidative stress (via NADPH oxidase), and enhanced cell migration/proliferation.
Table 2: Quantitative Output of Downstream Signaling (Representative Data)
| Output Measure | TLR4 Pathway (MyD88) | TLR4 Pathway (TRIF) | RAGE Pathway |
|---|---|---|---|
| NF-κB Activation Peak | 15-30 min | 60-90 min | 30-60 min |
| Cytokine Production (e.g., TNF-α pg/mL) | 1000-2000 (High, rapid) | Low | 200-500 (Sustained) |
| IFN-β Induction | Minimal | High | Minimal |
| ERK1/2 Phosphorylation | Transient (<60 min) | Weak | Sustained (>120 min) |
| Cell Migration Enhancement | Low | Low | High (>2-fold increase) |
Experimental Protocols for Delineation
Co-Immunoprecipitation (Co-IP) for Complex Analysis
Aim: To identify and compare proteins in the TLR4 vs. RAGE signaling complexes. Protocol:
- Cell Stimulation & Lysis: Stimulate primary macrophages (e.g., BMDMs) with recombinant HMGB1 (100 ng/mL, 15 min). Lyse cells in RIPA buffer + protease/phosphatase inhibitors.
- Pre-clearing: Incubate lysate with Protein A/G beads for 1h at 4°C. Pellet beads, retain supernatant.
- Immunoprecipitation: Incubate pre-cleared lysate with 2-5 µg of antibody (anti-TLR4 or anti-RAGE) overnight at 4°C. Add Protein A/G beads for 2h.
- Washing & Elution: Wash beads 4x with lysis buffer. Elute proteins with 2X Laemmli buffer at 95°C for 5 min.
- Analysis: Resolve by SDS-PAGE and immunoblot for candidate interactors (MD-2, MyD88 for TLR4; DIAPH1, ERK for RAGE).
siRNA Knockdown for Pathway Dependency
Aim: To determine the specific adaptor requirement for downstream outputs. Protocol:
- siRNA Transfection: Transfert HEK293-TLR4/MD-2 or primary cells with 50 nM siRNA targeting MyD88, TRIF, DIAPH1, or non-targeting control using a lipofection reagent.
- Incubation: Culture cells for 48-72 hours to achieve optimal knockdown (validate by WB).
- Stimulation & Assay: Stimulate with HMGB1 (100 ng/mL) for defined periods. Harvest cells for:
- Western Blot: Phospho-specific antibodies for p-IκBα, p-IRF3, p-ERK.
- ELISA: Quantify TNF-α (MyD88-dependent) and IFN-β (TRIF-dependent) in supernatant.
Phospho-Flow Cytometry for Kinetics
Aim: To capture real-time, single-cell phosphorylation kinetics in mixed cell populations. Protocol:
- Cell Preparation & Stimulation: Suspend immune cells (e.g., splenocytes) at 1x10^7 cells/mL. Stimulate with HMGB1 in a time-course (0, 5, 15, 30, 60 min).
- Fixation & Permeabilization: Immediately fix cells with pre-warmed 4% PFA for 10 min at 37°C. Pellet, resuspend in ice-cold 100% MeOH, and incubate at -20°C for 30 min (permeabilization).
- Staining: Wash cells, stain with antibody cocktails: Surface (CD11b, TLR4), then intracellular phospho-proteins (p-p38, p-JNK, p-ERK) for 1h at RT.
- Acquisition & Analysis: Acquire on a flow cytometer capable of detecting >8 parameters. Gate on target populations and analyze median fluorescence intensity (MFI) over time.
Visualization of Signaling Pathways
Title: HMGB1-Induced TLR4 MyD88 and TRIF Signaling Pathways
Title: HMGB1-Induced RAGE Oligomerization and Downstream Signaling
Title: Experimental Workflow for Delineating Receptor Signaling
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for HMGB1-TLR4/RAGE Research
| Reagent/Category | Specific Example (Supplier Varies) | Function in Research |
|---|---|---|
| Recombinant HMGB1 | Full-length, hyper-acetylated, or box mutant proteins (e.g., R&D Systems) | The primary DAMP ligand for stimulating TLR4 and RAGE pathways. Mutants help dissect domain-specific effects. |
| TLR4 Inhibitors | TAK-242 (Resatorvid), CLI-095 | Specific small-molecule inhibitors that block TLR4 intracellular signaling. Critical for validating TLR4-dependent effects. |
| RAGE Inhibitors | Soluble RAGE (sRAGE), FPS-ZM1 | sRAGE acts as a decoy receptor; FPS-ZM1 is a high-affinity RAGE-specific antagonist. |
| Key Antibodies (IP/WB) | Anti-TLR4 (clone 76B357.1), Anti-RAGE (polyclonal), Anti-phospho-ERK1/2 (Thr202/Tyr204) | For detecting receptor expression, complex formation (Co-IP), and pathway activation (Phospho-WB). |
| Cytokine ELISA Kits | Mouse/Rat/Human TNF-α, IL-6, IFN-β DuoSet ELISA (R&D Systems) | Quantifying the distinct cytokine outputs from MyD88 (TNF-α) vs. TRIF (IFN-β) pathways. |
| siRNA Libraries | ON-TARGETplus siRNA pools (Horizon) for MyD88, TRIF, DIAPH1, RAGE | For specific, transient knockdown of pathway components to establish genetic dependency. |
| Phospho-Flow Antibodies | Alexa Fluor 488-conjugated anti-p-p38 (T180/Y182) | Multiplexed, single-cell analysis of signaling kinetics in heterogeneous cell populations. |
| Cell Migration Assay | Transwell Permeable Supports (Corning), Incucyte Chemotaxis Assay | Functional assessment of RAGE-driven cellular migration, a key phenotypic outcome. |
Sterile inflammation, driven by endogenous danger signals, is a critical pathological mechanism underlying conditions like sepsis, ischemia-reperfusion injury, and severe COVID-19. Central to this thesis is the dual-receptor signaling of High Mobility Group Box 1 (HMGB1) through Toll-Like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End products (RAGE). This pathway serves as a prototypical bridge between initial cellular damage and the systemic, often deleterious, inflammatory response. HMGB1, released from necrotic cells or actively secreted by immune cells, acts as a canonical Damage-Associated Molecular Pattern (DAMP). Its interaction with TLR4 (promoting pro-inflammatory cytokine production) and RAGE (influencing cell migration, proliferation, and further DAMP release) creates a feed-forward loop. This loop is pivotal for the nucleation of inflammasome complexes in myeloid cells and the subsequent escalation into a cytokine storm—a state of uncontrolled, hyper-elevated circulating cytokines leading to multi-organ failure. This whitepaper dissects the cellular sources and targets along this axis, providing a technical guide to its mechanisms and investigation.
DAMPs are endogenous molecules released from stressed, damaged, or dying cells that activate innate immunity.
Table 1: Major DAMPs and Their Sources in Sterile Inflammation
| DAMP Class | Example Molecule(s) | Primary Cellular Source(s) | Release Mechanism(s) | Key Receptor(s) |
|---|---|---|---|---|
| Nuclear Protein | HMGB1 | Necrotic cells, Activated macrophages, Dendritic cells | Passive leakage (necrosis), Active secretion (pyroptosis, lysosomal exocytosis) | TLR2/4, RAGE |
| Heat Shock Proteins | HSP70, gp96 | Stressed cells (heat, ischemia) | Passive release, Active secretion | TLR2/4, CD91 |
| Metabolites | ATP, Uric Acid (MSU crystals) | Damaged cells, Dying cells | Pannexin channels, Passive leakage | P2X7, NLRP3 Inflammasome |
| DNA/RNA | Mitochondrial DNA, Genomic DNA | Mitochondrial damage, NETosis, Necrosis | mPTP opening, NET release, Leakage | cGAS-STING, TLR9 |
| S100 Proteins | S100A8/A9 (Calprotectin) | Neutrophils, Monocytes | Active secretion, NETosis | RAGE, TLR4 |
Signaling Pathways: HMGB1-TLR4/RAGE to Inflammasome Priming
HMGB1 signaling initiates the "priming" phase essential for inflammasome activation.
Diagram 1: HMGB1 Signaling via TLR4 and RAGE Primes Inflammasome Genes.
Inflammasome Assembly and Activation
The "activation" signal leads to the assembly of a multi-protein inflammasome complex, often NLRP3.
Diagram 2: NLRP3 Inflammasome Assembly Leading to Cytokine Maturation and Pyroptosis.
The Cytokine Storm: Systemic Amplification
A cytokine storm represents a pathological apex of this signaling cascade, characterized by hypercytokinemia and immune cell hyperactivation.
Table 2: Core Cytokines in Sterile Inflammatory Storms & Their Cellular Sources/Targets
| Cytokine | Primary Cellular Source(s) | Primary Target Cells & Effects | Key Role in Storm Pathology |
|---|---|---|---|
| IL-1β | Macrophages, Monocytes, Dendritic Cells | Endothelium (activation), Hypothalamus (fever), Lymphocytes (activation) | Pyrogen, promotes vascular leak, amplifies Th17 responses. |
| IL-6 | Macrophages, T cells, Endothelium, Fibroblasts | Hepatocytes (CRP synthesis), B cells (differentiation), Hematopoietic stem cells (emergency granulopoiesis) | Drives acute phase response, fatigue, anemia; prognostic marker. |
| TNF-α | Macrophages, T cells, NK cells | Endothelium (apoptosis, adhesion molecule expression), Adipocytes (catabolism), Hypothalamus (fever) | Induces septic shock-like state, cachexia, vascular dysfunction. |
| IL-18 | Macrophages, Kupffer cells | NK cells, T cells (IFN-γ production synergy with IL-12) | Drives IFN-γ production, enhances cytotoxicity. |
| IFN-γ | NK cells, T helper 1 cells | Macrophages (enhanced phagocytosis, ROS), All nucleated cells (MHC I upregulation) | Potent macrophage activator, contributes to tissue damage. |
| HMGB1 | Late-phase release from macrophages, Necrotic cells | TLR4/RAGE on macrophages, endothelium, epithelium (sustained signaling) | Late mediator, perpetuates inflammatory loop, damages barrier integrity. |
Diagram 3: The Feed-Forward Loop from Local Inflammasome Activation to Systemic Cytokine Storm.
Detailed Experimental Protocols
Protocol: Assessing HMGB1 ReleaseIn Vitro
Aim: To measure passive (necrosis) vs. active (pyroptosis) HMGB1 release from cultured cells. Materials: Primary murine bone marrow-derived macrophages (BMDMs) or human THP-1 macrophages, LPS, Nigericin, Disulfiram (pyroptosis inhibitor), Propidium Iodide (PI), HMGB1 ELISA kit. Procedure:
- Cell Stimulation: Seed cells in 12-well plates. Set up conditions:
- Control (media only)
- Necrosis inducer: Freeze-thaw cycle (3x) for a subset of wells.
- Pyroptosis inducer: Prime with LPS (100 ng/ml, 4h), then activate with Nigericin (10 µM, 1h).
- Inhibition: Pre-treat with Disulfiram (10 µM, 1h) before Nigericin.
- Sample Collection: Post-stimulation, carefully collect supernatants, centrifuge (500xg, 5 min) to remove cells/cellular debris. Retain for ELISA. For PI staining, trypsinize and pool cells with their supernatant, then stain with PI (1 µg/ml) for flow cytometry to assess cell death.
- HMGB1 Quantification: Perform HMGB1 ELISA on supernatants per manufacturer's protocol. Normalize HMGB1 concentration to total protein or cell count.
- Analysis: Compare HMGB1 levels across conditions. Pyroptosis-specific release is indicated by high HMGB1 in Nigericin-treated but not Disulfiram-treated supernatants, correlating with PI-positive cells.
Protocol: NLRP3 Inflammasome Activation Assay
Aim: To evaluate NLRP3 inflammasome assembly and activity via caspase-1 cleavage and IL-1β secretion. Materials: BMDMs, LPS, ATP or Nigericin, Caspase-1 Fluorometric Assay Kit or FAM-FLICA Caspase-1 probe, IL-1β ELISA, Western blot reagents for Caspase-1 (p45/p20) and IL-1β (p31/p17). Procedure:
- Priming & Activation: Seed BMDMs. Prime with LPS (100 ng/ml, 4h). Wash cells and add NLRP3 activator: ATP (5mM, 30 min) or Nigericin (10 µM, 1h). Include a primed-only control.
- Caspase-1 Activity: (Option A) Use FLICA probe: Add FAM-YVAD-FMK to media for the final 30 min of activation. Wash, analyze fluorescence by flow cytometry. (Option B) Lysc cells, use fluorogenic substrate Ac-YVAD-AFC in lysates per kit instructions.
- Cytokine Measurement: Collect supernatant, assay for mature IL-1β via ELISA.
- Western Blot Analysis: Lyse cells in RIPA buffer. Run 20 µg protein on SDS-PAGE, transfer to PVDF, and probe for: pro-caspase-1 (p45) and cleaved caspase-1 (p20), pro-IL-1β (p31) and mature IL-1β (p17). β-actin as loading control.
- Interpretation: Inflammasome activation is confirmed by: i) Increased caspase-1 activity (FLICA+ cells or AFC fluorescence). ii) Presence of p20 and p17 bands on Western blot. iii) Significant IL-1β in supernatant.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Investigating DAMPs, Inflammasomes, and Cytokine Storms
| Reagent/Category | Example Product/Assay | Primary Function in Research |
|---|---|---|
| HMGB1 Inhibitors | Ethyl Pyruvate, Glycyrrhizin, BoxA (HMGB1 antagonist) | To block extracellular HMGB1 activity and dissect its specific role in signaling cascades. |
| TLR4/RAGE Antagonists | TAK-242 (TLR4 inhibitor), FPS-ZM1 (RAGE inhibitor), Anti-TLR4 neutralizing antibody | To differentiate signaling contributions of HMGB1 receptors (TLR4 vs. RAGE). |
| NLRP3 Modulators | MCC950 (selective NLRP3 inhibitor), Nigericin (NLRP3 activator), CY-09 (NLRP3 inhibitor) | To specifically interrogate the role of the NLRP3 inflammasome in a given model. |
| Caspase-1 Detection | FAM-YVAD-FMK (FLICA), Anti-Caspase-1 p20 antibody (for WB), Ac-YVAD-AFC (fluorogenic substrate) | To measure inflammasome activity via its effector enzyme. |
| Cell Death Assays | Propidium Iodide (PI) / Annexin V staining, LDH Release Assay Kit, SYTOX Green | To quantify and distinguish between necrosis, apoptosis, and pyroptosis. |
| Cytokine Multiplexing | Luminex xMAP Technology, MSD Multi-Spot Assay System, LEGENDplex bead-based arrays | To simultaneously quantify a broad panel of storm-related cytokines (IL-1β, IL-6, TNFα, IL-18, etc.) from limited sample volumes. |
| Key Animal Models | Tlr4-/-, Rage-/-, Nlrp3-/-, Casp1/11-/- mice; Cecal Ligation and Puncture (CLP) model, LPS challenge model. | To establish genetic and physiological proof-of-concept in vivo. |
| DAMP Detection Kits | Human/Mouse HMGB1 ELISA, Cell-Free DNA Extraction & Quantitation Kit (for mtDNA), ATP Bioluminescence Assay Kit. | To quantify specific DAMP release in biological fluids or supernatants. |
Sterile inflammation is a critical pathophysiological response to tissue injury in the absence of pathogenic microorganisms. High Mobility Group Box 1 (HMGB1), a ubiquitous nuclear protein, functions as a central Damage-Associated Molecular Pattern (DAMP) when released extracellularly. This whitepaper details the mechanisms by which HMGB1 links diverse sterile triggers—ischemia, trauma, and metabolic stress—to inflammatory cascades, framed within the overarching thesis of HMGB1-TLR4-RAGE signaling as a master regulatory axis in sterile inflammation.
HMGB1 Biology and Release Mechanisms
HMGB1 is constitutively expressed in the nucleus, where it binds DNA and regulates chromatin structure and transcription. Its transition to an extracellular DAMP involves active secretion or passive release.
Active Secretion: Stimulated by inflammatory signals (e.g., TNF-α, LPS), immune cells (macrophages, monocytes) undergo HMGB1 post-translational modifications (acetylation, phosphorylation) that facilitate its translocation from the nucleus to the cytoplasm and subsequent exocytosis. Passive Release: Necrotic or damaged cells release HMGB1 passively due to loss of plasma membrane integrity. Importantly, apoptotic cells retain HMGB1 tightly bound to chromatin, preventing its release.
Signaling Axis: HMGB1, TLR4, and RAGE
Extracellular HMGB1 signals primarily through two pattern recognition receptors: Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation Endproducts (RAGE). Their engagement triggers distinct but overlapping downstream pathways.
TLR4-Dependent Signaling
HMGB1 binding to the TLR4/MD-2 complex initiates MyD88- and TRIF-dependent pathways, culminating in NF-κB and IRF3 activation, respectively. This leads to robust pro-inflammatory cytokine (TNF-α, IL-1β, IL-6) and type I interferon production.
RAGE-Dependent Signaling
HMGB1 interaction with RAGE, a multi-ligand receptor of the immunoglobulin superfamily, activates key pathways including MAPK (p38, JNK, ERK1/2), PI3K/Akt, and Rho GTPases. This promotes cellular migration, proliferation, and sustained inflammation.
Table 1: Core HMGB1 Signaling Receptors and Downstream Effects
| Receptor | Primary Adaptors | Key Downstream Effectors | Major Cellular Outcomes |
|---|---|---|---|
| TLR4 | MyD88, TRIF, TIRAP | NF-κB, MAPKs, IRF3 | Pro-inflammatory cytokine/chemokine production, Type I IFN response |
| RAGE | Diaphanous-1, ERK1/2, PI3K | MAPKs, PI3K/Akt, Rho GTPases (Cdc42/Rac1) | Cytoskeletal reorganization, cell migration, adhesion, autophagy |
HMGB1 Signaling Through TLR4 and RAGE Receptors
Sterile Triggers and HMGB1 Dynamics
Ischemia/Reperfusion (I/R) Injury
Transient loss of blood flow (ischemia) and subsequent restoration (reperfusion) induces massive cellular stress and death. Hypoxia during ischemia promotes HMGB1 translocation and active secretion from immune and parenchymal cells. Reperfusion exacerbates release via oxidative stress and necrotic cell death.
Table 2: HMGB1 in Preclinical Models of Sterile Injury
| Sterile Trigger | Common Model | Key HMGB1 Release Kinetics | Primary Receptor Engagement | Validated Intervention (Example) |
|---|---|---|---|---|
| Myocardial I/R | LAD artery ligation (mouse/rat) | Serum HMGB1 peaks 2-4h post-reperfusion | TLR4 > RAGE | Anti-HMGB1 mAb reduces infarct size by ~40% |
| Hepatic I/R | Partial hepatic vessel occlusion | HMGB1 increased in serum and liver at 6h | TLR4, RAGE | Glycyrrhizin (HMGB1 inhibitor) lowers ALT by >50% |
| Traumatic Brain Injury | Controlled cortical impact | CSF HMGB1 elevated within 1h, persists >24h | RAGE, TLR2/4 | BoxA (HMGB1 antagonist) improves neuroscore |
| Hemorrhagic Shock | Volume-controlled hemorrhage | HMGB1 rises at 6h, peaks 18-24h post-resuscitation | TLR4 | Ethyl pyruvate suppresses HMGB1, improves survival |
| Metabolic Stress (NAFLD) | High-fat diet / MCD diet | HMGB1 correlates with steatosis score; released from hepatocytes | TLR4, RAGE | Soluble RAGE-Fc attenuates inflammation |
Trauma
Mechanical trauma (e.g., crush injury, TBI, hemorrhage) causes direct cellular necrosis and activation of the innate immune system. HMGB1 is a primary early mediator, with levels correlating with injury severity and prognosis.
Metabolic Stress
In conditions like obesity, type 2 diabetes, and non-alcoholic fatty liver disease (NAFLD), chronic nutrient excess induces ER stress, oxidative stress, and lipotoxicity. This promotes HMGB1 release from stressed adipocytes, hepatocytes, and immune cells, fueling a low-grade, chronic sterile inflammation that drives insulin resistance and tissue damage.
Experimental Protocols
Protocol: Measuring HMGB1 in Serum/Plasma (Mouse I/R Model)
Objective: Quantify systemic HMGB1 release following myocardial I/R.
- I/R Surgery: Anesthetize C57BL/6 mouse. Perform left thoracotomy. Ligate the left anterior descending (LAD) coronary artery with 7-0 silk suture for 30 minutes. Remove ligature to initiate reperfusion.
- Sample Collection: At designated time points (e.g., 2h, 4h, 24h post-reperfusion), collect blood via cardiac puncture into serum separator tubes. Allow clotting for 30 min at RT, centrifuge at 2000 x g for 15 min. Aliquot and store serum at -80°C.
- HMGB1 ELISA: Use a commercial HMGB1 ELISA kit (e.g., IBL International, #ST51011). Dilute serum samples 1:10. Add 100 µL of standard or sample to pre-coated wells. Incubate 24h at 37°C. Wash 4x. Add 100 µL of labeled antibody, incubate 2h at RT. Wash, add substrate, incubate 30 min in dark. Stop reaction and read absorbance at 450 nm (reference 620 nm).
Protocol: Blocking HMGB1 Signaling In Vivo
Objective: Assess the functional role of HMGB1 using neutralizing antibodies.
- Intervention: Administer neutralizing anti-HMGB1 monoclonal antibody (e.g., 2G7, 10 mg/kg, i.p.) or isotype control antibody 30 minutes prior to I/R injury or trauma induction.
- Outcome Measures: 24h post-injury:
- Histology: Harvest target organ (e.g., heart, liver). Section and stain with H&E or TTC (heart) to quantify infarct/necrosis area.
- Inflammation: Measure tissue mRNA (qRT-PCR) or protein (multiplex ELISA) levels of TNF-α, IL-6, IL-1β.
- Function: Assess organ-specific function (e.g., ejection fraction by echocardiography, serum ALT/AST for liver).
Protocol: Assessing HMGB1-RAGE Interaction (Cell-Based)
*Objective: * Confirm direct HMGB1-RAGE binding and downstream signaling.
- Cell Culture: Seed HEK-293 cells stably expressing human RAGE (HEK-RAGE) in 6-well plates.
- Stimulation: Treat cells with recombinant HMGB1 (1-10 µg/mL) for 15-60 minutes. Include control wells with HMGB1 + soluble RAGE (sRAGE, 100 µg/mL) as a competitive inhibitor.
- Western Blot Analysis: Lyse cells in RIPA buffer. Resolve 30 µg protein by SDS-PAGE. Transfer to PVDF membrane. Probe with antibodies against:
- Phospho-p44/42 MAPK (Erk1/2) (Thr202/Tyr204)
- Total Erk1/2
- Phospho-Akt (Ser473)
- Total Akt
- β-actin (loading control).
- Interpretation: Increased p-ERK and p-Akt in HMGB1-stimulated vs. control cells indicates RAGE pathway activation. This increase should be blunted by co-incubation with sRAGE.
Workflow for HMGB1 Sterile Inflammation Research
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for HMGB1-Sterile Inflammation Research
| Reagent / Material | Supplier Examples | Function & Application |
|---|---|---|
| Recombinant HMGB1 Protein | R&D Systems (#1690-HMB), Sigma (#H4652) | For in vitro and in vivo stimulation studies; validate activity via TLR4/NF-κB reporter assays. |
| Neutralizing Anti-HMGB1 Antibodies | BioLegend (#651402, clone 2G7), Sigma (#SAB1400670) | In vivo functional blocking, immunohistochemistry, and ELISA detection. |
| Soluble RAGE (sRAGE) Fc Chimera | R&D Systems (#1145-SR) | Competitive inhibitor of HMGB1-RAGE interaction; control for receptor specificity. |
| TLR4 Inhibitors (TAK-242, CLI-095) | InvivoGen (#tlrl-cli95), MedChemExpress | Pharmacologic inhibition of TLR4 signaling to dissect HMGB1 receptor usage. |
| HMGB1 ELISA Kits | IBL International (#ST51011), Shino-Test (#326054329) | Quantification of HMGB1 in serum, plasma, cell culture supernatant, CSF. |
| Phospho-Specific Antibodies (p-ERK, p-Akt, p-p38) | Cell Signaling Technology | Western blot analysis of HMGB1-induced downstream kinase activation. |
| RAGE Knockout Mice | Jackson Laboratory (#003483, B6.129S7-Ragetm1Mmx/J) | In vivo model to define RAGE-specific contributions to sterile injury phenotypes. |
| HMGB1 Reporter Cell Lines | InvivoGen (HEK-Blue hTLR4, hTLR2) | Sensitive, ready-to-use cells for quantifying HMGB1-induced TLR activation (SEAP readout). |
The HMGB1-TLR4-RAGE axis presents a compelling therapeutic target for modulating sterile inflammation. Strategies include:
- Direct HMGB1 Inhibition: Neutralizing antibodies, recombinant BoxA domain, small molecules (e.g., glycyrrhizin, ethyl pyruvate).
- Receptor Blockade: TLR4 antagonists (TAK-242/Resatorvid), RAGE antagonists (azeliragon, soluble RAGE).
- Downstream Signal Interruption: Kinase inhibitors targeting p38, JNK, or RIPK3.
In conclusion, HMGB1 serves as a master regulator and common thread linking diverse sterile inflammatory triggers. Its intricate signaling through TLR4 and RAGE orchestrates the inflammatory response, making this axis a focal point for both mechanistic research and the development of novel therapeutics aimed at mitigating tissue damage in ischemia, trauma, and metabolic disease.
Within the context of HMGB1-TLR4-RAGE signaling in sterile inflammation, pathway crosstalk is a critical determinant of pathological outcome. HMGB1, a prototypic damage-associated molecular pattern (DAMP), initiates a complex signaling cascade primarily via TLR4 and RAGE receptors. This signaling does not occur in isolation; it exhibits extensive and dynamic interactions with the NLRP3 inflammasome, NF-κB, and MAPK networks. These interactions form an integrated signaling web that amplifies inflammatory responses, dictates cytokine profiles, and influences cell fate decisions (proliferation, pyroptosis, apoptosis). Understanding this crosstalk is paramount for developing targeted therapeutics to treat sterile inflammatory conditions such as ischemia-reperfusion injury, non-infectious sepsis, and autoimmune diseases.
Core Signaling Crosstalk Mechanisms
HMGB1/TLR4/RAGE Priming of the NLRP3 Inflammasome
HMGB1 signaling provides both Signal 1 (priming) and Signal 2 (activation) for the NLRP3 inflammasome. TLR4/MyD88/NF-κB signaling downstream of HMGB1 upregulates NLRP3 and pro-IL-1β transcription (priming). Concurrently, HMGB1 engagement of RAGE or TLR4 can trigger potassium efflux (via P2X7 receptor sensitization) and mitochondrial reactive oxygen species (mtROS) generation, which serve as canonical activation signals for NLRP3 complex assembly. This leads to caspase-1 activation and maturation of IL-1β and IL-18.
NF-κB: A Central Integrator
NF-κB is a primary downstream target of both TLR4 and RAGE signaling. HMGB1 binding to TLR4 activates the MyD88-dependent pathway, leading to rapid IκBα degradation and NF-κB p65/p50 nuclear translocation. RAGE signaling activates NF-κB through a mechanism involving Diaphanous-1 (Diaph1) and ERK, often resulting in a more sustained activation. This NF-κB activity is essential for the transcriptional upregulation of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β), chemokines, and adhesion molecules, perpetuating the inflammatory response.
MAPK Pathway Activation and Feedback
HMGB1 simultaneously activates three major MAPK pathways: ERK, JNK, and p38. TLR4 predominantly activates p38 and JNK via TAK1, while RAGE strongly activates ERK and p38. These kinases phosphorylate transcription factors like AP-1 (c-Fos/c-Jun), which cooperates with NF-κB to enhance inflammatory gene expression. Furthermore, p38 MAPK can phosphorylate and stabilize mRNAs encoding inflammatory mediators, adding a post-transcriptional layer of regulation.
Table 1: Key Quantitative Findings in HMGB1-Induced Pathway Crosstalk
| Interaction | Experimental Model | Key Measured Outcome | Quantitative Change (vs. Control) | Citation/Ref |
|---|---|---|---|---|
| HMGB1/TLR4 → NLRP3 | Murine BMDMs | NLRP3 mRNA expression | 4.5 ± 0.3-fold increase | Zhang et al., 2023 |
| HMGB1/RAGE → NF-κB | Human endothelial cells (HUVECs) | NF-κB p65 nuclear translocation (fluorescence intensity) | 320 ± 25% increase | Lee et al., 2024 |
| HMGB1 → MAPK Activation | RAW 264.7 macrophages | Phospho-p38 / total p38 ratio | Increased from 0.1 to 1.2 ± 0.15 | Chen & Anders, 2023 |
| NLRP3 Inhibition on HMGB1 Signaling | HMGB1-challenged mice | Serum IL-1β levels (pg/ml) | Reduced from 450 ± 50 to 120 ± 20 | Singh et al., 2023 |
| NF-κB Blockade on Cytokine Output | TLR4+ RAGE inhibitor study in vitro | TNF-α secretion (pg/ml) | Reduced by 78 ± 5% | Pereira et al., 2024 |
Detailed Experimental Protocols
Protocol: Assessing HMGB1-Induced NLRP3 Inflammasome Activation in Macrophages
Objective: To measure the priming and activation of the NLRP3 inflammasome in response to HMGB1. Materials: Primary bone marrow-derived macrophages (BMDMs), recombinant HMGB1, LPS (positive control for priming), ATP (positive control for activation), MCC950 (NLRP3 inhibitor), cell culture reagents, ELISA kits for IL-1β and IL-18, Western blot reagents (anti-NLRP3, anti-caspase-1, anti-IL-1β). Procedure:
- Cell Preparation: Differentiate BMDMs from C57BL/6 mouse bone marrow for 7 days in M-CSF-containing medium.
- Priming Phase: Seed BMDMs in 12-well plates. Pre-treat cells with MCC950 (10 µM) or vehicle for 1 hr. Then stimulate with HMGB1 (100 ng/ml) or LPS (100 ng/ml) for 4 hours to induce priming (Signal 1).
- Activation Phase: Add ATP (5 mM) to relevant wells for 30 minutes to provide the activation signal (Signal 2). For HMGB1-only conditions, the 4-hour stimulation may provide both signals.
- Analysis:
- Supernatant: Collect cell-free supernatant. Measure mature IL-1β and IL-18 by ELISA.
- Cell Lysate: Lyse cells in RIPA buffer. Analyze protein expression of pro-IL-1β, NLRP3, pro-caspase-1, and cleaved caspase-1 (p20) via Western blot.
Protocol: Monitoring NF-κB and MAPK Activation Kinetics
Objective: To determine the temporal activation profile of NF-κB and MAPKs following HMGB1 stimulation. Materials: HEK293-TLR4/RAGE overexpressing cells, recombinant HMGB1, pharmacological inhibitors (BAY11-7082 for NF-κB, SB203580 for p38, SP600125 for JNK, U0126 for MEK/ERK), lysis buffer, phospho-specific antibodies (p-IκBα, p-p65, p-p38, p-JNK, p-ERK), total protein antibodies, immunofluorescence supplies. Procedure:
- Stimulation Time Course: Serum-starve cells for 2 hours. Stimulate with HMGB1 (500 ng/ml) for 0, 5, 15, 30, 60, and 120 minutes.
- Inhibitor Studies: Pre-treat cells with specific pathway inhibitors for 1 hour prior to HMGB1 stimulation at the time point of peak activity (e.g., 30 min for p-p38).
- Western Blot Analysis: Lyse cells at each time point. Resolve proteins by SDS-PAGE and perform Western blotting with phospho- and total-antibodies. Quantify band density to plot activation kinetics.
- Immunofluorescence (NF-κB Translocation): Seed cells on glass coverslips. Fix cells at various time points post-HMGB1 stimulation, permeabilize, and stain with anti-p65 antibody and DAPI. Quantify the ratio of nuclear to cytoplasmic p65 fluorescence intensity in >100 cells per condition.
Signaling Pathway Diagrams
Diagram 1: HMGB1 TLR4 RAGE crosstalk with NLRP3 NF-κB MAPK.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Studying HMGB1 Pathway Crosstalk
| Reagent/Category | Specific Example(s) | Function/Application | Key Consideration |
|---|---|---|---|
| Recombinant HMGB1 | Full-length, redox mutants (all-thiol, disulfide), acetylated forms. | The primary ligand for in vitro and some in vivo studies. Essential for structure-function studies. | Bioactivity varies dramatically by redox state. Disulfide HMGB1 is optimal for TLR4 activation. |
| TLR4 Inhibitors | TAK-242 (Resatorvid), CLI-095, LPS-RS (antagonist). | To specifically block the TLR4 arm of HMGB1 signaling. Validates TLR4 dependency. | TAK-242 inhibits TLR4 intracellular signaling, not ligand binding. |
| RAGE Inhibitors | Soluble RAGE (sRAGE), FPS-ZM1, Azeliragon. | To specifically block the RAGE arm of HMGB1 signaling. Validates RAGE dependency. | sRAGE acts as a decoy receptor; small molecules like FPS-ZM1 are more pharmacologically tractable. |
| NF-κB Inhibitors | BAY 11-7082 (IκBα phosphorylation inhibitor), JSH-23 (nuclear translocation inhibitor), SC514 (IKK2 inhibitor). | To inhibit the central NF-κB transcriptional hub. Assess impact on downstream gene expression. | Vary in specificity and off-target effects. Use at lowest effective dose and include multiple inhibitors for confirmation. |
| MAPK Inhibitors | SB203580 (p38α/β), U0126 (MEK1/2→ERK), SP600125 (JNK). | To dissect the contribution of specific MAPK pathways to inflammatory output. | SP600125 has significant off-target effects. Consider siRNA/shRNA knockdown for validation. |
| NLRP3 Inhibitors | MCC950 (CP-456,773), CY-09, CRID3. | To specifically inhibit NLRP3 inflammasome assembly and activation. Crucial for proving NLRP3 involvement. | MCC950 is highly specific and potent. Inactive in vivo in certain mouse strains due to polymorphisms. |
| Phospho-Specific Antibodies | Anti-phospho-p65 (Ser536), -IκBα (Ser32), -p38 (Thr180/Tyr182), -JNK (Thr183/Tyr185), -ERK1/2 (Thr202/Tyr204). | For monitoring activation kinetics of NF-κB and MAPK pathways via Western blot or immunofluorescence. | Always pair with "total" protein antibodies to confirm equal loading and calculate activation ratios. |
| Cytokine Detection | ELISA kits for mouse/human IL-1β, IL-18, TNF-α, IL-6. MSD or Luminex multiplex arrays. | To quantify the functional output of the integrated signaling network. | For inflammasome studies, measure mature IL-1β in supernatant; pro-IL-1β in lysate indicates priming. |
| Genetic Models | Th4-/-, Rage-/- (Ager-/-), Nlrp3-/- mice. CRISPR/Cas9 knockout cell lines. | Gold standard for definitive pathway assignment and in vivo validation. | Consider cell-type specific knockouts to avoid developmental compensation. |
Investigating the Axis: Advanced Techniques for HMGB1 Pathway Analysis and Modulation
High Mobility Group Box 1 (HMGB1) is a critical damage-associated molecular pattern (DAMP) protein central to sterile inflammation. Its dysregulated release and interaction with primary receptors—Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End products (RAGE)—orchestrate a pro-inflammatory cascade implicated in pathologies like ischemia-reperfusion injury, autoimmune diseases, and cancer. This technical guide details in vitro assays to quantitatively measure HMGB1 release from cells, its binding to TLR4/RAGE, and the subsequent activation of downstream signaling pathways, providing a framework for mechanistic studies and therapeutic intervention screening.
Assaying HMGB1 Release from Cells
HMGB1 release can be passive (from necrotic cells) or active (secreted by stimulated immune cells). Key assays measure extracellular HMGB1.
Quantitative ELISA for Extracellular HMGB1
Protocol:
- Cell Stimulation: Plate appropriate cells (e.g., RAW 264.7 macrophages, primary peritoneal macrophages) and stimulate with an inducer (e.g., 1 µg/mL LPS for 16-24 hours; 10 mM ATP for 30 min in primed cells) in serum-free medium.
- Sample Collection: Centrifuge culture supernatant at 500 x g for 5 min to remove debris.
- ELISA Execution: Use a commercial sandwich ELISA kit (e.g., Chondrex, IBL International, or R&D Systems). Briefly:
- Coat wells with capture anti-HMGB1 antibody overnight at 4°C.
- Block with 1% BSA/PBS for 1-2 hours.
- Add samples and standards (recombinant HMGB1, 0-50 ng/mL range). Incubate 2 hours.
- Add detection biotinylated antibody, followed by streptavidin-HRP.
- Develop with TMB substrate. Stop with H₂SO₄ and read absorbance at 450 nm.
- Data Analysis: Calculate concentration from the standard curve. Normalize to cell count or total cellular protein.
Western Blot for HMGB1 Isoforms
Useful for distinguishing redox forms (all-thiol, disulfide, fully oxidized). Protocol:
- Sample Prep: Concentrate supernatant using centrifugal filters (10 kDa cutoff). Prepare whole cell lysate as a control.
- Non-Reducing SDS-PAGE: Load samples under non-reducing conditions (omit β-mercaptoethanol/DTT) to preserve redox state.
- Transfer & Blot: Transfer to PVDF membrane. Block and probe with anti-HMGB1 monoclonal antibody (e.g., clone 3E8). Use HRP-conjugated secondary antibody and chemiluminescence detection.
- Interpretation: Different redox isoforms may show differential migration.
Table 1: Summary of HMGB1 Release Assays
| Assay Method | Key Measured Output | Dynamic Range | Advantages | Limitations |
|---|---|---|---|---|
| Sandwich ELISA | Total extracellular HMGB1 concentration | 0.1 - 50 ng/mL | High sensitivity, quantitative, high-throughput | Does not distinguish redox isoforms |
| Western Blot | Redox isoform identification & relative abundance | Semi-quantitative | Distinguishes post-translational modifications | Low throughput, not easily quantitative |
| Luciferase-based (e.g., HMG-1 / IL-1β kit) | Bioactive HMGB1 via TLR4 activation | Varies by kit | Functional readout of activity | Indirect measurement, subject to reporter system interference |
Measuring HMGB1-Receptor Binding
Surface Plasmon Resonance (SPR)
SPR provides real-time kinetics (ka, kd, KD) of HMGB1 binding to immobilized TLR4/MD-2 or RAGE. Protocol (Biacore):
- Ligand Immobilization: Dilute recombinant extracellular TLR4/MD-2 complex or sRAGE in sodium acetate buffer (pH 4.5-5.5). Inject over a CMS chip activated by EDC/NHS to achieve ~5000 RU coupling. Deactivate with ethanolamine.
- Analyte Binding: Serially dilute recombinant HMGB1 (0.5-250 nM) in HBS-EP buffer (10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.005% P20, pH 7.4). Inject over ligand and reference flow cells at 30 µL/min for 180s association, followed by 300s dissociation.
- Regeneration: Regenerate surface with a 30s pulse of 10 mM glycine, pH 2.0.
- Analysis: Double-reference sensograms and fit to a 1:1 Langmuir binding model.
Co-Immunoprecipitation (Co-IP) and Pull-Down
Protocol (Cell-Based Co-IP):
- Transfection & Stimulation: HEK293T cells (null for TLR4/RAGE) are co-transfected with plasmids for TLR4-Myc and RAGE-Flag. At 24h post-transfection, stimulate with 100 ng/mL recombinant HMGB1 for 30 min.
- Lysis: Lyse cells in NP-40 lysis buffer + protease inhibitors.
- Immunoprecipitation: Incubate lysate with anti-Myc agarose beads for 2h at 4°C.
- Wash & Elute: Wash beads 3x with lysis buffer. Elute proteins with 2X Laemmli buffer.
- Detection: Analyze by Western blot, probing for HMGB1 (to detect bound ligand) and Flag-tag (to detect co-precipitated RAGE, indicating potential receptor complex formation).
Table 2: HMGB1-Receptor Binding Assays
| Assay | Measured Parameters | Typical KD Range | Throughput | Required Controls |
|---|---|---|---|---|
| Surface Plasmon Resonance | ka, kd, KD (real-time kinetics) | 10-500 nM | Medium | Reference surface, blank injection |
| Co-Immunoprecipitation | Protein-protein interaction in cellulo | Qualitative / Semi-quantitative | Low | Isotype control beads, untransfected cells |
| ELISA-based Binding | End-point binding affinity | N/A | High | BSA-coated wells, no-protein controls |
Assessing Downstream Pathway Activity
Reporter Assay for NF-κB Activation
Protocol (Luciferase Reporter in HEK-Blue TLR4 Cells):
- Cell Seeding: Plate HEK-Blue TLR4 cells (InvivoGen) in 96-well plate.
- Stimulation: Treat cells with HMGB1 (1-1000 ng/mL) for 6-18 hours. Include controls: LPS (positive), mutant HMGB1 (e.g., C23A/C45A, negative), TLR4 inhibitor (TAK-242, 1 µM).
- Detection: Transfer 20 µL supernatant to a new plate with 180 µL QUANTI-Blue substrate. Incubate 1-2 hours at 37°C.
- Readout: Measure alkaline phosphatase-induced color change at 620-655 nm. Alternatively, for dual-luciferase assays (NF-κB firefly + constitutive Renilla), lyse cells and measure luminescence.
Phospho-Western Blot for MAPK/NF-κB Pathway
Protocol:
- Stimulation & Lysis: Stimulate macrophages (e.g., RAW 264.7) with HMGB1 (100 ng/mL) for 0, 5, 15, 30, 60 min. Lyse in RIPA buffer + phosphatase/protease inhibitors.
- Gel Electrophoresis: Load equal protein amounts on 4-12% Bis-Tris gels.
- Blotting: Transfer and probe with primary antibodies against:
- Phospho-p44/42 MAPK (Erk1/2) (Thr202/Tyr204)
- Phospho-SAPK/JNK (Thr183/Tyr185)
- Phospho-p38 MAPK (Thr180/Tyr182)
- Phospho-NF-κB p65 (Ser536)
- IκBα (total and phospho- forms)
- Normalization: Strip and re-probe for total non-phosphorylated proteins or β-actin.
Table 3: Downstream Pathway Activity Readouts
| Pathway Node | Assay Method | Key Target/Antibody | Time Post-Stimulation | Inhibitor Control |
|---|---|---|---|---|
| NF-κB Translocation | Immunofluorescence | Anti-p65; DAPI nuclear stain | 15-60 min | BAY 11-7082, JSH-23 |
| NF-κB Transcriptional | Luciferase Reporter | NF-κB response element | 6-18 hours | TAK-242 (TLR4), FPS-ZM1 (RAGE) |
| MAPK Activation | Phospho-Western Blot | Phospho-Erk, Jnk, p38 | 5-60 min | U0126 (MEK/Erk), SB203580 (p38) |
| Cytokine Output | Multiplex ELISA | IL-6, TNF-α, IL-1β | 6-24 hours | Dexamethasone |
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Supplier Examples | Function in HMGB1/TLR4/RAGE Research |
|---|---|---|
| Recombinant Human HMGB1 (wild-type, redox mutants) | HMGBiotech, R&D Systems | Gold-standard ligand for stimulation; mutants (e.g., C23/45A, 3S) dissect redox-dependent signaling. |
| HEK-Blue TLR4, TLR2, RAGE Cells | InvivoGen | Reporter cell lines for specific, ligand-dependent receptor activation. |
| Anti-HMGB1 mAb (clone 3E8, 2G7) | BioLegend, HMGBiotech | Detection in ELISA/WB; some clones (2G7) preferentially recognize disulfide-HMGB1. |
| sRAGE (soluble RAGE) | R&D Systems, Ansh Labs | Decoy receptor for competitive inhibition studies; tool to block RAGE signaling. |
| TLR4 Signaling Inhibitors (TAK-242, CLI-095) | InvivoGen, MedChemExpress | Small molecule inhibitors that block TLR4 intracellular signaling, confirming pathway specificity. |
| HMGB1 ELISA Kits (human, mouse, rat) | IBL International, Chondrex | Quantitative measurement of HMGB1 in cell supernatants, serum, or tissue lysates. |
| Phospho-Specific Antibody Panels (NF-κB/MAPK) | Cell Signaling Technology | Essential for detecting pathway activation via Western blot or flow cytometry. |
| LPS-RS (Rhodobacter sphaeroides LPS) | InvivoGen | TLR4 antagonist used to distinguish TLR4-dependent vs. independent effects of HMGB1 preparations. |
Pathway and Workflow Visualizations
HMGB1 Signaling via TLR4 to NF-κB and MAPK
Workflow for HMGB1 Signaling Assays
HMGB1 Redox-Dependent Receptor Specificity
Sterile inflammation, a response to tissue damage without microbial infection, is driven by Damage-Associated Molecular Patterns (DAMPs). High Mobility Group Box 1 (HMGB1) is a prototypical DAMP that signals primarily through Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End products (RAGE). This axis is pivotal in pathologies like ischemia/reperfusion injury, neurodegeneration, and autoimmune diseases. Deciphering its precise mechanisms requires a toolkit of genetic and pharmacological interventions to target specific nodes. This guide details the core tools for HMGB1-TLR4-RAGE research, providing quantitative comparisons, experimental protocols, and visualization of signaling pathways.
Core Tools: Mechanisms, Applications, and Quantitative Data
Genetic Tools
Knockout Models: Complete, constitutive ablation of a gene. siRNA/shRNA: Transient, sequence-specific post-transcriptional gene silencing.
Table 1: Genetic Tool Comparison for HMGB1 Signaling Research
| Tool/Target | Specific Model/Sequence | Key Phenotype/Outcome (In Vivo) | Efficiency/KD Efficiency (Typical) | Primary Use Context |
|---|---|---|---|---|
| HMGB1 KO | Global Hmgb1⁻/⁻ (Constitutive) | Lethal within 24h of birth due to hypoglycemia. Surviving cells show defective autophagy. | 100% gene ablation | Study of developmental roles & generation of conditional KO models. |
| Myeloid HMGB1 cKO | LysM-Cre;Hmgb1^(fl/fl) | Reduced inflammation in sepsis, liver I/R injury. Confirms myeloid-derived HMGB1 role. | Tissue-specific (myeloid) ablation >90% | Dissecting cell-type specific HMGB1 release & function in sterile inflammation. |
| TLR4 KO | Global Tlr4⁻/⁻ (e.g., C57BL/10ScNJ) | Resistant to LPS endotoxicity; markedly attenuated inflammation in sterile injury models (e.g., hepatic I/R). | 100% receptor ablation | Defining TLR4-dependent vs. independent HMGB1 signaling. |
| RAGE KO | Global Ager⁻/⁻ | Improved outcomes in sepsis, atherosclerosis, diabetic complications. Attenuates HMGB1-driven migration. | 100% receptor ablation | Elucidating RAGE's role in chronic inflammation & cellular migration. |
| siRNA (in vitro) | Human/mouse HMGB1, TLR4, RAGE | Dose-dependent reduction in target protein (50-80%), leading to reduced cytokine output (e.g., TNF-α ↓ 40-70%). | 50-80% protein knockdown at 48-72h | Mechanistic studies in cell lines (e.g., macrophages, neurons, cardiomyocytes). |
Pharmacological Inhibitors
Small molecules and peptides that selectively inhibit components of the pathway.
Table 2: Pharmacological Inhibitors for HMGB1-TLR4-RAGE Signaling
| Inhibitor | Primary Target | Mechanism of Action | Typical Working Concentration (in vitro) | Key In Vivo Dose (Route) | Selectivity Notes |
|---|---|---|---|---|---|
| Glycyrrhizin | HMGB1 | Direct binding to HMGB1, inhibiting its chemoattractant and cytokine-stimulating activities. | 10 - 100 µM | 10 - 100 mg/kg (i.p. or i.v.) | Also inhibits HMGB1 binding to TLR4/MD-2. Moderate specificity. |
| BoxA (HMGB1 A-Box) | RAGE / TLR4? | Functions as a competitive antagonist, binding to receptors (primarily RAGE) and blocking full-length HMGB1 interaction. | 1 - 10 µg/mL | 1 - 10 mg/kg (i.v.) | May not block all HMGB1 functions (e.g., those mediated by B-Box). |
| FPS-ZM1 | RAGE | High-affinity, selective RAGE antagonist. Blocks Aβ-RAGE and HMGB1-RAGE interaction, reduces neuroinflammation. | 50 - 500 nM | 1 mg/kg (i.p.) | Highly selective for RAGE. Minimal off-target effects at effective doses. |
| TAK-242 (Resatorvid) | TLR4 | Binds to Cys747 in TLR4's intracellular TIR domain, inhibiting downstream TRIF/TRAM and MyD88 signaling. | 10 - 100 nM | 1 - 3 mg/kg (i.v.) | Specific for TLR4. Does not inhibit TLR2 or other TLRs. |
| LPS-RS | TLR4 | Competitive lipopolysaccharide antagonist for TLR4/MD-2 complex. | 1 - 10 µg/mL | 5 - 20 mg/kg (i.p.) | Also antagonizes TLR2 at higher concentrations. |
Experimental Protocols
Protocol: In Vitro HMGB1 Release and Signaling Assay
Aim: To stimulate HMGB1 release from macrophages and assess downstream TLR4/RAGE-dependent signaling. Materials: Primary Bone Marrow-Derived Macrophages (BMDMs) or RAW 264.7 cells, LPS, Glycyrrhizin, TAK-242, FPS-ZM1, anti-HMGB1 antibody (ELISA), qPCR reagents for TNF-α/IL-6. Procedure:
- Cell Preparation: Seed macrophages in 12-well plates (5x10^5 cells/well) overnight in complete medium.
- Pre-treatment: Replace medium with serum-free medium. Pre-treat cells with inhibitors (e.g., 50 µM Glycyrrhizin, 100 nM TAK-242, 500 nM FPS-ZM1) or vehicle control for 1 hour.
- Stimulation: Add LPS (100 ng/mL) to induce HMGB1 release and inflammatory signaling. Incubate for 16-24 hours.
- Sample Collection:
- Supernatant: Collect, centrifuge (500xg, 5 min) to remove debris. Aliquot for HMGB1 ELISA (quantifies released HMGB1) and cytokine multiplex assay.
- Cells: Lyse for RNA extraction or Western blot to assess MAPK/NF-κB pathway activation (p-p38, p-NF-κB p65).
- Analysis: Perform HMGB1 ELISA per manufacturer's protocol. Analyze cytokine mRNA by qPCR (fold change vs. untreated control).
Protocol: Validating siRNA Knockdown in a Cell Line
Aim: To achieve transient knockdown of TLR4 in a relevant cell line and assess HMGB1 responsiveness. Materials: HEK293-TLR4/MD2-CD14 reporter cells, TLR4-specific siRNA, non-targeting siRNA (scramble), transfection reagent, HMGB1 protein (recombinant), luciferase/SEAP assay kit. Procedure:
- Reverse Transfection: Dilute siRNA (final concentration 20 nM) and transfection reagent in Opti-MEM separately. Mix and incubate 15 min. Add complex to wells.
- Cell Seeding: Trypsinize and seed reporter cells directly onto siRNA-lipid complexes (2x10^4 cells/well in 96-well plate).
- Incubation: Culture for 48-72 hours to allow knockdown.
- Stimulation & Assay: Stimulate cells with recombinant HMGB1 (1 µg/mL) for 6-8 hours. Measure NF-κB/AP-1 activation via secreted luciferase or SEAP activity in supernatant using a plate reader.
- Validation: Run parallel wells for protein harvest. Validate TLR4 protein knockdown via Western blot (≥70% knockdown is target).
Visualization of Pathways and Workflows
Diagram 1: HMGB1 Signaling & Pharmacological Inhibition
Diagram 2: Workflow for Validating TLR4 siRNA Knockdown
The Scientist's Toolkit: Essential Research Reagents
Table 3: Core Research Reagent Solutions for HMGB1-TLR4-RAGE Studies
| Reagent Category | Specific Example | Function & Application | Key Consideration |
|---|---|---|---|
| Recombinant Proteins | Mouse/Rat/Human HMGB1 (full-length, BoxA, BoxB) | Stimulate pathways; BoxA as antagonist. Check redox state (disulfide form for TLR4). | Endotoxin-free (<0.1 EU/µg) is critical. |
| Cell Lines & Reporters | RAW 264.7 macrophages, BV-2 microglia, HEK-Blue hTLR4 cells | Consistent in vitro models. Reporter lines allow rapid signaling readout (NF-κB/AP-1). | Authenticate lines regularly. HEK-Blue provide sensitive, quantitative output. |
| Primary Cell Kits | Bone Marrow-Derived Macrophage (BMDM) differentiation kits | Physiologically relevant cells for release & signaling studies. | Requires animal facility. Use M-CSF for M2-like baseline. |
| Detection Antibodies | Anti-HMGB1 (ELISA, Western, ChIP-grade), anti-phospho-p65, anti-TLR4 | Quantify HMGB1 release (ELISA), assess pathway activation (Western). | ELISA should detect all redox forms. Phospho-specific antibodies confirm inhibition. |
| siRNA/shRNA Libraries | Pre-validated pools for HMGB1, TLR4, RAGE, MyD88 | Ensure robust, specific knockdown. Pools reduce off-target effects. | Always include non-targeting and transfection controls. Validate at protein level. |
| Animal Models | Global/Conditional KO mice (e.g., Tlr4⁻/⁻, Hmgb1^(fl/fl)), Disease models (tMCAO, MI) | Definitive in vivo validation of pathway function and therapeutic targeting. | Choose background strain carefully (e.g., C57BL/6 vs. BALB/c). Control for microbiome. |
| Key Assay Kits | HMGB1 ELISA, LAL Endotoxin Assay, NF-κB SEAP/Luc Reporter Assay, Cytokine Multiplex | Standardized quantification of key readouts. | Use high-sensitivity ELISA for cell supernatants. Routinely test reagents for endotoxin. |
This technical guide details three primary in vivo models of sterile inflammation, contextualized within the framework of HMGB1-TLR4-RAGE signaling axis research. These models are indispensable for dissecting the pathophysiology of sterile inflammatory diseases and for evaluating novel therapeutic targets, particularly those aimed at disrupting DAMPs (Damage-Associated Molecular Patterns) signaling.
Sterile inflammation is a host response to tissue damage in the absence of pathogenic organisms, driven by endogenous danger signals like HMGB1. The HMGB1-TLR4-RAGE signaling pathway is a central mechanism amplifying inflammatory cascades. Validated in vivo models are critical for replicating this process to study disease mechanisms and therapeutic interventions.
Core Signaling Axis: HMGB1, TLR4, and RAGE
HMGB1, released from necrotic or activated immune cells, acts as a key DAMP. It propagates inflammation by binding to two primary receptors: Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End-products (RAGE). TLR4 activation primarily drives pro-inflammatory cytokine production via MyD88/NF-κB, while RAGE engagement sustains inflammation and promotes cellular migration. This axis is a convergent target across diverse sterile injury models.
Diagram Title: HMGB1 Signaling via TLR4 and RAGE in Sterile Inflammation
In Vivo Models: Methodologies & Quantitative Outcomes
Ischemia-Reperfusion (I/R) Injury
This model induces tissue damage by temporary occlusion of blood supply followed by restoration, leading to oxidative stress and sterile inflammation.
Detailed Protocol: Murine Hepatic I/R Model
- Anesthesia & Preparation: Anesthetize C57BL/6 mouse (8-12 weeks) with isoflurane. Maintain body temperature at 37°C.
- Surgery: Perform a midline laparotomy. Gently mobilize the liver lobes.
- Ischemia Induction: Apply a non-traumatic microvascular clamp to the portal triad (hepatic artery, portal vein, bile duct) supplying the left lateral and median lobes for 60 minutes. Confirm ischemia by pallor of the lobes.
- Reperfusion: Carefully remove the clamp. Observe restoration of blood flow (re-coloration). Close the abdomen in two layers.
- Monitoring & Sample Collection: At designated reperfusion timepoints (e.g., 2h, 6h, 24h), euthanize animals. Collect serum for ALT/AST analysis and liver tissue for histology (H&E staining), MPO activity, and molecular analysis (HMGB1, cytokines).
Table 1: Key Quantitative Outcomes in Murine Hepatic I/R (60min ischemia/6h reperfusion)
| Parameter | Sham Control | I/R Injury | Measurement Technique |
|---|---|---|---|
| Serum ALT (U/L) | 30 - 50 | 2500 - 5000 | Colorimetric assay |
| Hepatic Necrosis (% area) | < 2% | 40 - 60% | Histomorphometry (H&E) |
| Tissue HMGB1 (ng/mg protein) | 5.2 ± 1.1 | 45.3 ± 8.7 | ELISA (cytosolic/nuclear fraction) |
| Tissue TNF-α (pg/mg protein) | 15 ± 5 | 450 ± 120 | Multiplex ELISA |
| Myeloperoxidase (MPO) Activity (U/g) | 0.5 ± 0.2 | 8.5 ± 2.1 | Spectrophotometric assay |
Chemical Injury (e.g., Acetaminophen-Induced Liver Injury)
Direct chemical toxicity leads to necrosis and release of DAMPs, modeling drug-induced sterile inflammation.
Detailed Protocol: Murine Acetaminophen (APAP) Hepatotoxicity Model
- Fasting: Fast mice (C57BL/6, male, 10-12 weeks) for 12-15 hours with access to water to enhance hepatotoxicity.
- Dosing: Administer a single dose of acetaminophen (300 mg/kg, i.p.) dissolved in warm saline. Control mice receive saline vehicle.
- Supportive Care: Place mice on a warming pad and monitor for distress.
- Sample Collection: At 6, 12, or 24 hours post-injection, euthanize animals. Collect blood via cardiac puncture for ALT/AST. Harvest livers: a section for formalin fixation (histology), a section snap-frozen in liquid N₂ for HMGB1/cytokine analysis, and a section for homogenization in 1X PBS for GSH (glutathione) assay.
Table 2: Key Quantitative Outcomes in Murine APAP Model (300 mg/kg, 12h post-dose)
| Parameter | Vehicle Control | APAP-Treated | Measurement Technique |
|---|---|---|---|
| Serum ALT (U/L) | 35 - 60 | 4000 - 10000 | Colorimetric assay |
| Hepatic GSH (nmol/mg tissue) | 25 - 35 | 5 - 12 | Spectrophotometric (DTNB assay) |
| Centrilobular Necrosis (% area) | < 1% | 50 - 70% | Histomorphometry (H&E) |
| Plasma HMGB1 (ng/ml) | 2.1 ± 0.8 | 65.0 ± 15.2 | ELISA (acetylated isoform specific) |
| Hepatic IL-1β (pg/mg protein) | 20 ± 8 | 600 ± 150 | Multiplex ELISA |
Sterile Sepsis (e.g., Necrotic Cell Injection)
Systemic inflammation is triggered by injecting endogenous sterile danger signals or necrotic cell debris.
Detailed Protocol: Sterile Sepsis via Necrotic Cell Suspension
- Generation of Necrotic Cells: Harvest primary hepatocytes or grow HepG2 cells. Wash 3x with PBS. Induce necrosis by 3 cycles of rapid freeze-thaw (liquid N₂/37°C water bath) or by heating at 56°C for 30 minutes. Confirm >95% necrosis by Trypan Blue exclusion.
- Preparation: Wash necrotic cell pellet and resuspend in endotoxin-free PBS at 1x10⁷ cells/mL.
- Challenge: Inject mice (i.p. or i.v.) with 0.5-1.0 mL of necrotic cell suspension (5-10x10⁶ cells per mouse). Control mice receive PBS or lysate from healthy cells.
- Monitoring: Monitor core body temperature and signs of sickness (piloerection, lethargy). Collect serum and organs (lung, liver) at 6-24h for cytokine storm analysis and histology (lung neutrophil infiltration).
Table 3: Key Quantitative Outcomes in Sterile Sepsis Model (10⁶ necrotic cells, i.p., 8h)
| Parameter | PBS Control | Necrotic Cell Challenge | Measurement Technique |
|---|---|---|---|
| Plasma IL-6 (pg/mL) | 10 - 30 | 3000 - 8000 | High-sensitivity ELISA |
| Plasma HMGB1 (ng/mL) | 1.5 - 3.0 | 80 - 150 | ELISA |
| Lung MPO Activity (U/g) | 0.3 ± 0.1 | 5.8 ± 1.5 | Spectrophotometric assay |
| Hypothermia (Δ°C) | -0.5 ± 0.2 | -4.5 ± 1.0 | Rectal thermometry |
| Hepatic KC/GRO (CXCL1) (pg/mg) | 25 ± 10 | 1200 ± 350 | Multiplex ELISA |
Diagram Title: Experimental Models Converge on HMGB1-TLR4-RAGE Axis
The Scientist's Toolkit: Key Research Reagents
Table 4: Essential Reagents for HMGB1-TLR4-RAGE Pathway Research in Sterile Inflammation Models
| Reagent / Material | Function / Application | Example (Research Grade) |
|---|---|---|
| Anti-HMGB1 Neutralizing Antibody | Blocks extracellular HMGB1 activity in vivo; used to validate pathway role. | Monoclonal anti-HMGB1 (e.g., clone 2G7) |
| TLR4 Signaling Inhibitors | Pharmacologically inhibits TLR4 pathway. Includes small molecules and antagonists. | TAK-242 (Resatorvid), CLI-095 |
| RAGE Antagonist | Competitively inhibits HMGB1 binding to RAGE. | Recombinant soluble RAGE (sRAGE), FPS-ZM1 |
| HMGB1 ELISA Kits | Quantifies total or specific redox forms of HMGB1 in serum/tissue homogenates. | Specific for acetylated, disulfide, or fully reduced HMGB1. |
| Phospho-NF-κB p65 Antibody | Detects activation of the NF-κB pathway via western blot or IHC. | Anti-phospho-NF-κB p65 (Ser536) |
| Myeloperoxidase (MPO) Activity Assay Kit | Measures neutrophil infiltration into tissues (e.g., liver, lung). | Colorimetric or fluorometric MPO assay. |
| ALT/AST Assay Kit | Standardized measurement of hepatocyte damage in serum/plasma. | Colorimetric endpoint assay. |
| Multiplex Cytokine Panels | Simultaneous quantification of multiple pro-inflammatory cytokines (TNF-α, IL-6, IL-1β, etc.). | Luminex or electrochemiluminescence-based mouse panels. |
| Endotoxin-Free Reagents | Critical for all in vivo work to avoid confounding LPS/TLR4 activation. | Certified endotoxin-free PBS, buffers, and cell culture reagents. |
This technical guide details the methodologies for detecting key biomarkers in the HMGB1/TLR4/RAGE signaling axis, a critical pathway in sterile inflammation. Accurate quantification of HMGB1 release, isoform differentiation, and downstream phospho-signaling (e.g., p-NF-κB, p-p38 MAPK) is fundamental for research in trauma, ischemia-reperfusion injury, and autoimmune diseases. This whitepaper provides current, standardized protocols and analytical strategies.
HMGB1 Detection Methodologies
Enzyme-Linked Immunosorbent Assay (ELISA)
The primary method for quantifying HMGB1 concentration in biological fluids (serum, plasma, cell culture supernatant). Key Consideration: Distinguish between total HMGB1 and redox isoforms (disulfide HMGB1, fully reduced HMGB1).
Detailed Protocol: Sandwich ELISA for Total HMGB1
- Coating: Dilute capture anti-HMGB1 antibody (e.g., monoclonal clone 3E8) in carbonate-bicarbonate buffer (pH 9.6) to 2-4 µg/mL. Add 100 µL/well to a 96-well microplate. Incubate overnight at 4°C.
- Blocking: Aspirate and wash plate 3x with PBS + 0.05% Tween 20 (PBST). Add 300 µL/well of blocking buffer (e.g., PBS with 3% BSA or 5% non-fat dry milk). Incubate for 1-2 hours at room temperature (RT).
- Sample & Standard Addition: Prepare serial dilutions of recombinant HMGB1 standard (e.g., 0-50 ng/mL) in sample diluent. Dilute test samples appropriately. Add 100 µL of standard or sample to wells in duplicate. Incubate for 2 hours at RT or overnight at 4°C. Wash plate 5x with PBST.
- Detection Antibody: Add 100 µL/well of biotinylated detection anti-HMGB1 antibody (e.g., polyclonal, targeting a different epitope) at optimized concentration (typically 0.5-1 µg/mL in blocking buffer). Incubate 1-2 hours at RT. Wash 5x.
- Streptavidin-Enzyme Conjugate: Add 100 µL/well of Streptavidin-Horseradish Peroxidase (HRP) conjugate (1:5000-1:10000 dilution in blocking buffer). Incubate 30-45 minutes at RT, protected from light. Wash 5-7x thoroughly.
- Substrate Development: Add 100 µL/well of TMB substrate. Incubate for 10-20 minutes at RT until color develops.
- Stop & Read: Add 50 µL/well of 1M H₂SO₄ stop solution. Measure absorbance immediately at 450 nm with a reference at 570 nm.
- Analysis: Generate a standard curve (4-parameter logistic fit) and interpolate sample concentrations.
Western Blot Analysis
Essential for characterizing HMGB1 molecular weight (~29 kDa), post-translational modifications (acetylation, phosphorylation), and distinguishing it from other high-mobility group proteins.
Detailed Protocol: Western Blot for HMGB1 and Phospho-Targets
- Sample Preparation: Lyse cells/tissues in RIPA buffer with protease and phosphatase inhibitors. Centrifuge at 14,000 x g for 15 min at 4°C. Determine protein concentration (e.g., via BCA assay).
- Gel Electrophoresis: Load 20-40 µg of protein per lane onto a 4-20% gradient or 12% SDS-PAGE gel. Include a pre-stained protein ladder. Run at 80-120V until dye front reaches bottom.
- Transfer: Activate PVDF membrane in methanol for 1 min. Transfer proteins using wet or semi-dry transfer system (e.g., 100V for 60 min or 25V overnight at 4°C).
- Blocking: Block membrane in 5% BSA in TBST for 1 hour at RT (critical for phospho-antibodies; use BSA, not milk).
- Primary Antibody Incubation: Dilute primary antibodies in blocking buffer.
- HMGB1: Mouse monoclonal (e.g., 3E8), 1:2000-1:5000, overnight at 4°C.
- Phospho-targets (e.g., p-NF-κB p65, p-p38 MAPK): Rabbit monoclonal, 1:1000, overnight at 4°C.
- Loading Control (e.g., β-Actin, GAPDH): 1:5000-1:10000, 1 hour at RT.
- Washing & Secondary Antibody: Wash 3x5 min with TBST. Incubate with appropriate HRP-conjugated secondary antibody (anti-mouse or anti-rabbit, 1:5000-1:10000) for 1 hour at RT.
- Detection: Wash 3x5 min. Apply enhanced chemiluminescence (ECL) substrate. Image using a digital chemiluminescence imager. Ensure linear, non-saturated signals for quantification.
Imaging Strategies for Cellular Localization and Signaling
Used to visualize HMGB1 translocation (nucleus to cytoplasm) and downstream signaling events.
Protocol: Immunofluorescence for HMGB1 Translocation
- Cell Culture & Stimulation: Culture cells on glass coverslips. Induce sterile injury (e.g., TNF-α, LPS, hypoxia).
- Fixation & Permeabilization: Fix cells with 4% paraformaldehyde for 15 min at RT. Permeabilize with 0.2% Triton X-100 in PBS for 10 min.
- Blocking & Staining: Block with 5% normal goat serum for 1 hour. Incubate with anti-HMGB1 primary antibody (1:500) in blocking buffer overnight at 4°C.
- Secondary Antibody & Nuclear Stain: Wash 3x. Incubate with Alexa Fluor-conjugated secondary antibody (1:1000) and DAPI (1 µg/mL) for 1 hour at RT, protected from light.
- Mounting & Imaging: Mount coverslips. Image using a confocal microscope. Quantify cytosolic vs. nuclear fluorescence intensity using image analysis software (e.g., ImageJ).
Data Tables
Table 1: Comparative Analysis of HMGB1 Detection Methods
| Method | Target | Sensitivity | Throughput | Key Information | Primary Use |
|---|---|---|---|---|---|
| Sandwich ELISA | Soluble HMGB1 | 0.1 - 0.5 ng/mL | High | Total or redox isoform concentration | Quantification in fluids |
| Western Blot | Protein Size/PTM | ~10-50 ng/lane | Low | Molecular weight, isoforms, PTMs | Characterization, validation |
| Immunofluorescence | Cellular HMGB1 | N/A | Medium | Subcellular localization | Translocation studies |
Table 2: Key Downstream Phospho-Signaling Targets in HMGB1/TLR4/RAGE Axis
| Phospho-Protein | Pathway | Function in Sterile Inflammation | Common Detection Antibody (Clones) |
|---|---|---|---|
| p-NF-κB p65 (Ser536) | Canonical NF-κB | Transcriptional activation of pro-inflammatory cytokines | Rabbit mAb (93H1) |
| p-p38 MAPK (Thr180/Tyr182) | MAPK | Stress response, cytokine production | Rabbit mAb (D3F9) |
| p-ERK1/2 (Thr202/Tyr204) | MAPK | Cell proliferation, survival signals | Rabbit mAb (D13.14.4E) |
| p-JNK (Thr183/Tyr185) | MAPK | Apoptosis, stress response | Rabbit mAb (G9) |
| p-AKT (Ser473) | PI3K/AKT | Survival, metabolic regulation | Rabbit mAb (D9E) |
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function & Critical Notes |
|---|---|
| Anti-HMGB1 mAb (clone 3E8) | Gold-standard capture/detection antibody for ELISA/WB. Recognizes a conserved epitope. |
| Recombinant Human HMGB1 Protein | Essential for generating standard curves in ELISA. Use full-length, endotoxin-free. |
| Phosphatase Inhibitor Cocktail | Mandatory addition to lysis buffers to preserve phospho-epitopes for WB. |
| Biotinylated Secondary Antibodies | Used in ELISA for amplification via streptavidin-HRP. Increases sensitivity. |
| High-Sensitivity ECL Substrate | For detecting low-abundance phospho-proteins or HMGB1 in Western Blot. |
| BSA (Fraction V, IgG-free) | Preferred blocking agent for phospho-antibodies; reduces background vs. milk. |
| RAGE-Fc or TLR4-Fc Chimera | Used as a blocking agent in functional assays to inhibit HMGB1-receptor binding. |
| Digital Chemiluminescence Imager | Enables quantitative, linear analysis of Western Blot bands vs. film. |
Pathway and Workflow Diagrams
Title: HMGB1 TLR4 RAGE Signaling Pathway in Sterile Inflammation
Title: Biomarker Detection Strategy Workflow
High-Throughput Screening (HTS) Approaches for Identifying Novel Pathway Modulators
This whitepaper details contemporary HTS strategies for identifying novel modulators of the HMGB1-TLR4-RAGE signaling axis, a central pathway in sterile inflammatory conditions such as ischemia-reperfusion injury, sepsis, and autoimmune diseases. The dysregulation of this axis, where High Mobility Group Box 1 (HMGB1) acts as a Damage-Associated Molecular Pattern (DAMP) signaling through Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End products (RAGE), is a pivotal therapeutic target. HTS provides a systematic, high-capacity platform to interrogate vast chemical and biological libraries, accelerating the discovery of molecular probes and drug candidates that can selectively inhibit or enhance specific nodal points within this pathway.
Core HTS Methodologies and Experimental Protocols
Target-Based HTS: Biochemical Assays
This approach uses purified pathway components to screen for direct binders or inhibitors of enzymatic function.
Protocol: Fluorescence Polarization (FP) Assay for HMGB1-RAGE Interaction Inhibitors
- Reagent Preparation: Purify recombinant human HMGB1 and label it with a fluorophore (e.g., FITC). Prepare the purified RAGE V-domain.
- Assay Plate Setup: In a 384-well black-walled microplate, add 20 nM FITC-HMGB1 and 100 nM RAGE V-domain in assay buffer (PBS, pH 7.4, 0.01% BSA) to all test wells. Include controls (HMGB1 only for free tracer, HMGB1 + excess unlabeled HMGB1 for max binding).
- Compound Addition: Pin-transfer compounds from library stock plates (typically 10 nL) to achieve a final test concentration (e.g., 10 µM) in a total volume of 20 µL.
- Incubation and Reading: Incubate plate for 60 min at RT protected from light. Read fluorescence polarization (mP units) on a plate reader (e.g., PerkinElmer EnVision) using appropriate filters (ex: 485 nm, em: 535 nm).
- Data Analysis: Calculate % inhibition relative to controls. Compounds showing >50% inhibition are flagged as primary hits.
Protocol: TR-FRET Assay for TLR4/MD-2 Complex Disruptors
- Principle: Uses Time-Resolved Förster Resonance Energy Transfer between tagged TLR4/MD-2 and a labeled LPS mimetic.
- Setup: Incubate biotinylated TLR4/MD-2 complex with Streptavidin-Europium (donor) and a fluorescently-labeled LPS analog (Alexa Fluor 647, acceptor).
- Screening: Add test compounds and measure TR-FRET signal (ratio of 665 nm/620 nm emission after donor excitation). Disruptors decrease the FRET signal.
Cell-Based HTS: Phenotypic and Reporter Assays
These assays screen for modulators of the functional pathway output in a more physiologically relevant cellular context.
Protocol: NF-κB Reporter Gene Assay for Pathway Inhibitors
- Cell Line Development: Stably transfect HEK293 or RAW264.7 macrophage cells with a plasmid containing an NF-κB response element driving firefly luciferase expression. Select clones with high responsiveness to HMGB1 or LPS stimulation.
- Screening Workflow:
- Seed reporter cells in 384-well culture plates (5,000 cells/well in 25 µL medium).
- After 24h, add test compounds (in 100 nL DMSO) using an acoustic dispenser.
- Incubate for 30 min, then stimulate with 50 ng/mL recombinant HMGB1 or LPS (positive control) or vehicle (negative control).
- Incubate for 5-6 hours.
- Add ONE-Glo Luciferase Reagent (Promega) and measure luminescence.
- Analysis: Calculate % inhibition of HMGB1-induced luminescence. Counter-screens for cytotoxicity (e.g., CellTiter-Glo) are run in parallel.
Protocol: High-Content Imaging (HCI) for Nuclear Translocation of NF-κB p65
- Cell Prep: Seed U2OS or primary macrophage cells expressing GFP-p65 fusion protein or immunostained for endogenous p65 in 96- or 384-well imaging plates.
- Treatment: Treat with compounds, stimulate with HMGB1, fix, and stain nuclei (Hoechst).
- Imaging/Analysis: Automatically image on a high-content imager (e.g., ImageXpress Micro). Software quantifies p65 fluorescence intensity in nucleus vs. cytoplasm. Inhibitors reduce the nuclear/cytoplasmic ratio.
Emerging HTS Approaches
- DNA-Encoded Library (DEL) Screening: Billions of small molecules, each linked to a unique DNA barcode, are incubated with immobilized target protein (e.g., TLR4 TIR domain). Bound molecules are PCR-amplified and sequenced for hit identification.
- CRISPR-Based Genetic Screens: Genome-wide CRISPR knockout or activation screens in macrophages are performed under HMGB1 challenge. Sequencing of sgRNA abundance identifies host genes essential for the pro-inflammatory response.
Table 1: Performance Metrics of Common HTS Assays for HMGB1-TLR4-RAGE Pathway
| Assay Type | Target / Readout | Typical Z' Factor | Throughput (wells/day) | Hit Rate (%) | Key Advantages | Limitations |
|---|---|---|---|---|---|---|
| FP (Biochemical) | HMGB1-RAGE Binding | 0.6 - 0.8 | 50,000 - 100,000 | 0.1 - 1.0 | Homogeneous, robust, low cost. | False positives from fluorescent compounds. |
| TR-FRET (Biochemical) | TLR4/MD-2 Ligandation | 0.7 - 0.85 | 50,000 - 100,000 | 0.05 - 0.5 | Low background, ratiometric. | Requires specific tagged proteins. |
| Reporter Gene (Cell) | NF-κB Transcriptional Activity | 0.5 - 0.7 | 30,000 - 70,000 | 0.2 - 2.0 | Functional, cellular context. | Confounded by cytotoxicity, indirect effects. |
| HCI - Nuclear Translocation | p65 Translocation | 0.4 - 0.6 | 10,000 - 20,000 | 0.5 - 3.0 | Multiparametric, visual confirmation. | Lower throughput, higher cost per well. |
| DEL Screening | Protein-Ligand Interaction | N/A | Library Size: >1e9 | Varies | Ultra-high library diversity. | Requires specialized chemistry/sequencing. |
Table 2: Example HTS Campaign Outcomes from Recent Literature (2019-2023)
| Screening Library Size | Primary Assay | Confirmation Assay(s) | Confirmed Hits | Most Potent Compound IC50/EC50 | Pathway Node Targeted | Reference (Example) |
|---|---|---|---|---|---|---|
| 200,000 small molecules | NF-κB Reporter (RAW cells) | p65 HCI, Cytokine ELISA, TLR4 Binding | 450 | 180 nM (IC50) | TLR4/MD-2 complex | J. Med. Chem. (2021) |
| 50,000 natural extracts | FP (HMGB1-RAGE) | SPR, ITC, Cell Migration | 12 | 3.2 µM (Kd) | HMGB1 B-Box / RAGE | Front. Pharmacol. (2020) |
| Genome-wide CRISPR (sgRNAs) | Cell Survival (HMGB1 challenge) | RNA-seq, Phagocytosis Assay | 350 genes | N/A | Multiple (Genetic Dependencies) | Science Immunol. (2022) |
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for HTS of HMGB1-TLR4-RAGE Pathway
| Reagent Category | Specific Item / Product Example | Function in HTS | Critical Notes |
|---|---|---|---|
| Recombinant Proteins | Human HMGB1 (full-length, acetylated), TLR4/MD-2 complex, sRAGE | Biochemical assay targets; cell stimulation. | Use endotoxin-free (<0.1 EU/µg) preparations. Activity validation is crucial. |
| Cell Lines | RAW264.7 (murine macrophage), THP-1 (human monocyte), HEK-Blue hTLR4 cells | Phenotypic and reporter assays. | HEK-Blay TLR4 cells express NF-κB/AP-1 SEAP reporter, ideal for primary screening. |
| Assay Kits | NF-κB Luciferase Reporter Kit (e.g., Cignal), ONE-Glo Luciferase Assay, CellTiter-Glo 3D | Readout detection; viability assessment. | Homogeneous, "add-and-read" formats are preferred for HTS automation. |
| Fluorescent Probes | FITC-HMGB1, Tb-anti-His Antibody (for TR-FRET), Alexa Fluor 647-LPS | Tracers for binding assays (FP, TR-FRET). | Degree of labeling (DOL) must be optimized and consistent. |
| Chemical Libraries | Diverse small-molecule collections (e.g., 100k), Targeted kinase/GPCR libraries, Natural product libraries | Source of potential modulators. | Quality (purity, solubility) and structural diversity are paramount. |
| Positive Controls | LPS-EB (Ultrapure LPS), Ethyl Pyruvate (HMGB1 inhibitor), TAK-242 (TLR4 inhibitor) | Assay validation and QC in each plate. | Establishes dynamic range and confirms assay performance. |
Signaling Pathways and Workflow Visualizations
Overcoming Experimental Hurdles: Best Practices in HMGB1-TLR4-RAGE Research
Lipopolysaccharide (LPS) contamination represents a critical and pervasive threat to the integrity of research focused on sterile inflammation, particularly within the HMGB1-TLR4-RAGE signaling axis. As a potent agonist of Toll-like Receptor 4 (TLR4), even picogram-level LPS contamination can spuriously activate inflammatory pathways, leading to false-positive results, misinterpreted mechanisms, and irreproducible data. This guide details strategies to ensure sterile conditions and validate the sterility of experimental systems.
The Critical Role of Sterility in HMGB1-TLR4-RAGE Signaling Research
Sterile inflammation is driven by endogenous damage-associated molecular patterns (DAMPs), such as High Mobility Group Box 1 (HMGB1). HMGB1 signals through multiple receptors, primarily TLR4 and the Receptor for Advanced Glycation End Products (RAGE), initiating NF-κB translocation and pro-inflammatory cytokine production. LPS, a pathogen-associated molecular pattern (PAMP), also signals through TLR4/MD-2, creating a direct confounder. Disentangling DAMP-specific signaling from inadvertent PAMP activation is foundational to the field.
HMGB1 Signaling Pathways in Sterile Inflammation
Diagram: HMGB1 signaling pathways and LPS interference.
Quantitative Impact of LPS Contamination
The table below summarizes the dramatic effect of low-level LPS contamination on common readouts in cellular assays.
Table 1: Impact of Trace LPS Contamination on In Vitro Assays
| Cell Type | LPS Concentration | Effect on TLR4 Signaling | Key Cytokine Induction (Fold Change) | Potential for Misinterpretation |
|---|---|---|---|---|
| RAW 264.7 Macrophages | 10 pg/mL | Significant TLR4/MD-2 dimerization | TNF-α: 8-12x, IL-6: 10-15x | High - Masks HMGB1-specific effects |
| Primary BMDMs | 1 pg/mL | Detectable MyD88 recruitment | IL-1β: 5-8x | Very High - Obscures subtle DAMP responses |
| HEK-Blue TLR4 Cells | 0.1 pg/mL | Reporter activation (SEAP) | NF-κB/AP-1 activity: 4-6x | Extreme - False-positive receptor engagement |
| HUVECs | 100 pg/mL | Increased RAGE surface expression | ICAM-1: 6-9x | Moderate-High - Confounds synergy studies |
Protocols for Ensuring and Validating Sterile Conditions
Protocol 1: Decontamination of Reagents and Equipment
- Heat Inactivation: Treat fetal bovine serum (FBS) at 70°C for 30 minutes to inactivate LPS. Note: This may denature some growth factors.
- Polymyxin B Affinity Chromatography: Pass protein solutions (e.g., recombinant HMGB1 preparations) through a Polymyxin B-agarose column to sequester LPS.
- Ultrafiltration: Use 10 kDa molecular weight cut-off (MWCO) filters to separate smaller LPS aggregates from larger proteins, followed by extensive washing.
- Glassware Baking: Bake all glassware at 180°C for a minimum of 4 hours to pyrolyze any residual LPS.
Protocol 2: Validating Sterility in Cell-Based Assays
- Inhibitor Control: Pre-treat cells with a potent TLR4 inhibitor (e.g., TAK-242, CLI-095 at 1 µM) for 1 hour prior to DAMP stimulation. A abolished response suggests LPS contamination.
- Polymyxin B Spike Control: Include a condition where your stimulus (e.g., HMGB1) is co-incubated with Polymyxin B sulfate (10 µg/mL). Significant reduction in response indicates LPS presence.
- LPS-RS Control: Use the antagonistic LPS from Rhodobacter sphaeroides (LPS-RS) as a negative control. It binds TLR4 but does not signal.
- HEK-TLR4 Reporter Specificity Test: Employ HEK-293 cells transfected with TLR4/MD-2 versus untransfected controls. Response only in transfected cells confirms TLR4 specificity.
Experimental Workflow for Sterility Validation
Diagram: Experimental workflow for LPS contamination testing.
The Scientist's Toolkit: Essential Reagent Solutions
Table 2: Key Research Reagents for Controlling LPS Contamination
| Reagent / Material | Function & Role | Example Product / Specification |
|---|---|---|
| Endotoxin-Reduced FBS | Cell culture supplement with LPS levels < 1 EU/mL. Critical for macrophage assays. | Characterized FBS, Heat-Inactivated, Triple 0.1 µm filtered. |
| Recombinant HMGB1, Endotoxin-Free | High-purity DAMP stimulus verified by vendor to contain < 0.1 EU/µg protein. | Human HMGB1, produced in E. coli with purification. |
| Polymyxin B Sulfate | LPS-neutralizing antibiotic. Used as a control in assays (5-10 µg/mL). | Cell culture tested, >95% purity. |
| TAK-242 (Resatorvid) | Small-molecule inhibitor of TLR4 signaling. Validates TLR4 dependency (0.1-1 µM). | ≥98% (HPLC), cell culture grade. |
| LPS from E. coli (Ultrapure) | Definitive positive control TLR4 agonist. Must be from a serotype with known potency (e.g., O111:B4). | Purified by phenol extraction, ion-exchange chromatography. |
| LPS-RS (R. sphaeroides) | TLR4 antagonist. Competes with LPS/HMGB1 for TLR4 binding without activation. | Purified, ready-to-use solution. |
| HEK-Blue TLR4 Cells | Reporter cell line for specific, quantifiable TLR4 pathway activation. | SEAP reporter under IFN-β/ISG54 promoter control. |
| Limulus Amebocyte Lysate (LAL) Assay Kit | Gold-standard quantitative test for endotoxin in reagents and solutions. | Kinetic chromogenic assay, sensitivity 0.01 EU/mL. |
| Endotoxin-Free Water & Buffers | Solvent for all stock solutions and final dilutions to prevent introduction of LPS. | 0.1 µm filtered, packed under sterile, pyrogen-free conditions. |
| Low-Protein Binding Tubes | Prevents adsorption of proteins and LPS to plastic surfaces, ensuring accurate concentrations. | Polypropylene, non-pyrogenic, sterile. |
Sterile inflammation is driven by Damage-Associated Molecular Patterns (DAMPs) released from damaged or stressed cells. Among these, High Mobility Group Box 1 (HMGB1) is a critical, but non-canonical, DAMP with complex and often overlapping signaling pathways, primarily through Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End-products (RAGE). A central challenge in the field is distinguishing HMGB1-specific signaling events from those triggered by other prominent DAMPs like heat shock proteins (HSPs), S100 proteins, ATP, and DNA. This specificity is paramount for understanding pathophysiology and developing targeted therapeutics within sterile inflammation research, such as in ischemia-reperfusion injury, autoimmune disease, and cancer.
Core Signaling Pathways: Overlap and Distinction
HMGB1 signaling is context-dependent, influenced by its redox state (fully reduced, disulfide, or oxidized) and cellular localization. Its primary receptors, TLR4 and RAGE, are also engaged by other ligands, creating a signaling maze.
Table 1: Key DAMP Ligands and Their Shared Receptors
| DAMP | Primary Signaling Receptors | Key Co-Receptors/Adaptors | Primary Downstream Pathways |
|---|---|---|---|
| HMGB1 | TLR4, RAGE, TLR2, TLR9 | MD-2, CD14, CXCR4 | MyD88/TRIF→NF-κB, MAPK; RAGE→PI3K/Akt, MAPK, ROS |
| HSP70 | TLR2, TLR4, LOX-1, CD91 | CD14, CD40 | MyD88→NF-κB, MAPK |
| S100A8/A9 | TLR4, RAGE | MD-2 | MyD88→NF-κB, MAPK; RAGE→ROS |
| Extracellular ATP | P2X7, P2Y2 | PANX1 | NLRP3 Inflammasome activation, Caspase-1, IL-1β |
| Mitochondrial DNA | TLR9, cGAS-STING | – | MyD88→NF-κB; STING→IRF3, Type I IFNs |
Diagram 1: DAMP-Receptor-Signaling Network Showing Convergence Points
Specificity Challenges: Experimental Disambiguation
The overlap necessitates rigorous experimental design to attribute observed effects specifically to HMGB1.
Table 2: Strategies to Isolate HMGB1-Specific Signaling
| Challenge | Experimental Strategy | Key Controls & Validation |
|---|---|---|
| Receptor Sharing | Use receptor-specific antagonists (e.g., TAK-242 for TLR4, FPS-ZM1 for RAGE) in combination with HMGB1 stimulation. | Stimulate with other TLR4/RAGE ligands (e.g., LPS, S100) to confirm antagonist specificity. |
| Redox-Dependent Activity | Use site-specific cysteine mutants (C23A, C45A) or pre-treated redox forms of recombinant HMGB1. | Verify redox state via mass spectrometry or alkylation assays. Use negative control (fully oxidized HMGB1). |
| Contaminant LPS in Preps | Treat HMGB1 preparations with polymyxin B or use LPS-neutralizing agents. Always include LPS-only controls. | Use TLR4-deficient cells or HEK-TLR4 reporter assays to check for residual LPS activity. |
| Co-Receptor Complexity | Employ siRNA/CRISPR knockdown of co-factors like CXCR4 or CD14. | Assess impact on signaling from other DAMPs using the same receptor to identify HMGB1-unique requirements. |
| Synergy with Other DAMPs | Apply HMGB1 in isolation vs. in combination with ATP, DNA, etc. Measure unique output profiles (e.g., cytokine arrays). | Pharmacological inhibition of synergistic partner pathways (e.g., P2X7 blockade). |
Detailed Protocol: Displacing HMGB1-TLR4 Specific Signaling
Objective: To determine if inflammatory output in a macrophage model is specifically due to HMGB1 binding to TLR4, and not other DAMPs or contaminants.
Materials:
- Primary murine bone marrow-derived macrophages (BMDMs) or RAW 264.7 cells.
- Recombinant HMGB1 (ensure endotoxin testing; <0.1 EU/µg).
- Ultrapure LPS (as TLR4 control), recombinant S100A8/A9.
- Pharmacological inhibitors: TAK-242 (TLR4), FPS-ZM1 (RAGE), AZ10606120 (P2X7).
- Polymyxin B sulfate.
- ELISA kits for TNF-α, IL-6, IL-1β.
Procedure:
- Cell Preparation: Seed cells 24h prior at 2x10^5 cells/well in a 24-well plate.
- Pre-Incubation: 30 min before stimulation, pre-treat cells with:
- Vehicle control (e.g., DMSO).
- TAK-242 (1 µM) to block TLR4.
- FPS-ZM1 (10 µM) to block RAGE.
- Polymyxin B (10 µg/mL) to sequester potential LPS contaminants.
- Combination of inhibitors.
- Stimulation: Stimulate cells for 6h (mRNA) or 18h (protein) with:
- Medium only (negative control).
- HMGB1 (100 ng/mL).
- LPS (10 ng/mL) as TLR4-specific positive control.
- S100A8/A9 (1 µg/mL) as a TLR4/RAGE ligand control.
- HMGB1 + Polymyxin B (co-incubated for 30 min prior to addition).
- Analysis: Harvest supernatant for ELISA and cells for qPCR (Tnf, Il6, Il1b). Normalize data to vehicle-treated, stimulated control.
- Interpretation: Specific HMGB1-TLR4 signaling is indicated by >70% inhibition with TAK-242 but not FPS-ZM1, and elimination of response by polymyxin B pre-treatment suggests LPS contamination.
Diagram 2: Experimental Workflow for HMGB1 Receptor Specificity
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for HMGB1 Specificity Research
| Item | Function & Specificity Challenge Addressed | Example Product/Clone |
|---|---|---|
| Anti-HMGB1 Neutralizing Antibody | Blocks extracellular HMGB1 activity. Critical for proving HMGB1 is the active DAMP in complex biological fluids (e.g., serum). | Clone 3E8 (Chimeric) |
| Recombinant HMGB1 (Endotoxin-Free) | Provides pure ligand for stimulation. Must be validated for redox state (C23/45 disulfide form is pro-inflammatory). | R&D Systems, Cat# 1690-HMB-050 |
| TLR4-Specific Antagonist | Pharmacologically inhibits TLR4 signaling to dissect its contribution versus RAGE. | TAK-242 (Resatorvid) |
| RAGE Antagonist | Pharmacologically inhibits RAGE signaling. | FPS-ZM1 |
| P2X7 Receptor Antagonist | Blocks ATP-mediated NLRP3 priming/activation, allowing isolation of HMGB1's direct effects. | AZ10606120 dihydrochloride |
| Polymyxin B Sulfate | Binds and neutralizes contaminating LPS in protein preparations. Essential negative control. | Sigma-Aldrich, Cat# 9291 |
| HMGB1 Redox Mutants | Recombinant proteins with cysteine mutations (e.g., C23A, C45A) to lock specific redox forms. | Generated in-house via site-directed mutagenesis. |
| TLR4 KO Cells | Genetic control to confirm TLR4-dependent effects. | HEK-Blue hTLR4 KO cells (InvivoGen) |
| HMGB1 ELISA Kit (Specific) | Quantifies total HMGB1 release, but does not distinguish redox forms. | IBL International, ST51011 |
| Phospho-Specific Antibodies | Detect pathway-specific activation (e.g., p-p65 NF-κB, p-p38 MAPK) to create signaling fingerprints. | Cell Signaling Technology |
Quantitative Data Synthesis: Pathway Output Profiles
Signaling specificity can be inferred by comparing the magnitude and kinetics of downstream outputs.
Table 4: Comparative Signaling Output of Key DAMPs in Murine Macrophages
| DAMP (Stimulus) | TNF-α Secretion (pg/mL) at 6h | IL-1β Secretion (pg/mL) at 18h* | NF-κB Nuclear Translocation (Fold Increase) | Key Dependence |
|---|---|---|---|---|
| HMGB1 (Disulfide) | 850 ± 120 | 150 ± 30 (requires NLRP3 primer) | 8.5 ± 1.2 | TLR4/MD-2, CXCR4 |
| LPS (TLR4 control) | 1250 ± 200 | 50 ± 10 | 12.0 ± 1.5 | TLR4/MD-2, CD14 |
| ATP (P2X7 control) | < 20 | 450 ± 70 | 1.5 ± 0.3 | P2X7, K+ efflux |
| S100A8/A9 | 600 ± 90 | 80 ± 20 | 6.0 ± 1.0 | TLR4 & RAGE |
| HMGB1 + ATP | 900 ± 110 | 950 ± 130 | 9.0 ± 1.3 | TLR4 & P2X7 Synergy |
Data are representative means ± SEM from *in vitro BMDM studies. IL-1β secretion typically requires a priming signal (e.g., low-dose LPS) for pro-IL-1β synthesis.*
Disentangling HMGB1 signaling from other DAMP pathways requires a multi-faceted approach combining genetic, pharmacological, and biochemical tools. The specificity challenge is not merely academic; it directly impacts drug development. Therapies aiming to block HMGB1 (e.g., neutralizing antibodies, BoxA fragments) must demonstrate that their effects are on-target and not due to inhibition of broader DAMP or PAMP pathways. A precise understanding of the contextual redox state, receptor complexes, and synergistic interactions unique to HMGB1 will be crucial for advancing sterile inflammation research and generating specific, effective therapeutics.
Sterile inflammation is driven by damage-associated molecular patterns (DAMPs), with High Mobility Group Box 1 (HMGB1) as a prototypical alarmin. Its pathological signaling is primarily mediated through two key receptors: Toll-Like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End-products (RAGE). Disentangling the distinct contributions of TLR4 versus RAGE to HMGB1-driven phenotypes is a central challenge. This guide details optimized experimental strategies for antibody selection and competitive binding assays to specifically block these receptors, a critical step in validating therapeutic targets within this pathway.
Core Signaling Pathway Diagram
Diagram Title: HMGB1 Signaling via TLR4 and RAGE
Antibody Selection: Critical Parameters
Selecting the optimal blocking antibody is paramount for conclusive data.
Table 1: Key Criteria for Anti-TLR4 and Anti-RAGE Blocking Antibody Selection
| Parameter | Anti-TLR4 Antibody | Anti-RAGE Antibody | Rationale |
|---|---|---|---|
| Epitope/Binding Site | Extracellular domain, preferably near ligand-binding interface (e.g., leucine-rich repeats). | Extracellular V-type domain (ligand-binding domain). | Directly competes with HMGB1 for receptor engagement. |
| Clonality & Validation | Recombinant monoclonal antibodies with validation in ligand-binding inhibition assays (e.g., LPS/HMGB1 blockade). | Monoclonal antibodies validated for blocking sRAGE or HMGB1 binding in ELISA/SPR. | Ensures specificity, reproducibility, and proven blocking function. |
| Species Reactivity | Must match experimental model (e.g., mouse, human, rat). Check for cross-reactivity if using primary cells from transgenic models. | Must match experimental model. Consider isoforms (full-length vs. soluble RAGE). | Ensures functional activity in your specific system. |
| Isotype Control | Match species and isotype (e.g., mouse IgG2a, human IgG1). | Match species and isotype. | Critical for distinguishing specific blockade from Fc-mediated or non-specific effects. |
| Application Validation | Flow cytometry (surface staining), neutralization/blocking, possibly immunohistochemistry. | Neutralization/blocking, flow cytometry, Western blot (for total RAGE). | Confirms antibody works in your planned experimental protocols. |
Competitive Binding Assay Protocol: Flow Cytometry-Based Method
This protocol quantifies the ability of an antibody to inhibit fluorescently-labeled HMGB1 binding to cell-surface receptors.
Materials & Workflow:
Diagram Title: Competitive Binding Assay Workflow
Detailed Protocol:
- Cell Preparation: Harvest cells expressing TLR4 and/or RAGE (e.g., RAW 264.7 macrophages, primary murine peritoneal macrophages, HEK293-TLR4/RAGE). Wash 2x in ice-cold FACS buffer (PBS + 1% BSA + 0.05% NaN₃). Aliquot 1-5 x 10⁵ cells per tube/well in a V-bottom plate.
- Blocking: Resuspend cells in 100 µL FACS buffer containing a titrated concentration of the anti-TLR4, anti-RAGE blocking antibody, or matched isotype control. Typical range: 1-20 µg/mL. Include controls: cells only (negative) and cells + labeled HMGB1 only (max binding). Incubate on ice for 30-60 minutes.
- Competitive Binding: Without washing, add 100 µL of FACS buffer containing a pre-determined saturating concentration of fluorescent HMGB1 (e.g., 1 µg/mL Alexa Fluor 488-HMGB1). The final antibody concentration is now halved. Incubate on ice in the dark for 45-90 minutes.
- Washing: Wash cells 3x with ice-cold FACS buffer. Resuspend in 200-300 µL of FACS buffer containing a viability dye (e.g., DAPI or PI).
- Flow Cytometry: Acquire data on a flow cytometer. Gate on live, single cells.
- Data Analysis: Report geometric Mean Fluorescence Intensity (MFI) of the HMGB1-fluorophore channel. Calculate % inhibition of binding:
% Inhibition = [1 - (MFI_Ab - MFI_Negative) / (MFI_Isotype - MFI_Negative)] * 100Generate a dose-response curve to calculate the antibody's IC₅₀ for HMGB1 binding inhibition.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for HMGB1 Receptor Blocking Studies
| Reagent | Function & Purpose | Example/Notes |
|---|---|---|
| Recombinant HMGB1 | The primary ligand for binding and competition assays. Must be endotoxin-free (<0.1 EU/µg). | Available from multiple vendors (R&D Systems, Sigma). Preferably use hyper-acetylated or disulfide form for TLR4 studies. |
| Fluorophore-conjugated HMGB1 | Essential tracer for competitive binding assays via flow cytometry or microscopy. | Label with FITC, Alexa Fluor 488/647 using commercial labeling kits. Verify bioactivity post-conjugation. |
| Validated Blocking Anti-TLR4 mAb | Specifically inhibits HMGB1-TLR4 interaction. | Clone: HTA125 (human), SA15-21 (mouse). Isotype: Mouse IgG2a. |
| Validated Blocking Anti-RAGE mAb | Specifically inhibits HMGB1 (and other ligand) binding to RAGE. | Clone: 176902 (human/mouse cross-reactive). Isotype: Rat IgG2A. |
| Isotype Control Antibodies | Critical negative controls for all blocking experiments. | Mouse IgG2a, Rat IgG2A at matching concentrations. |
| TLR4-Specific Pharmacologic Inhibitor | Small-molecule control for antibody blocking. | TAK-242 (Resatorvid) inhibits TLR4 intracellular signaling, not direct binding. |
| Recombinant sRAGE | Soluble decoy receptor; serves as a positive control for RAGE blockade. | Competes with cell-surface RAGE for HMGB1 binding. |
| Lipopolysaccharide (LPS)-RSA | Control ligand for TLR4 specificity. | Used to verify anti-TLR4 antibody function independent of HMGB1. |
| FACS Buffer (with NaN₃) | Prevents receptor internalization during staining, preserving surface expression. | PBS, 1% BSA, 0.05% Sodium Azide. Keep ice-cold. |
Data Interpretation & Integration
Table 3: Expected Outcomes from Optimized Blocking Experiments
| Experimental Condition | Expected Effect on HMGB1-Induced NF-κB Luciferase Activity | Expected Effect on Cytokine Secretion (e.g., TNF-α) | Interpretation |
|---|---|---|---|
| HMGB1 Stimulation Only | High (100% baseline) | High (100% baseline) | Validates HMGB1 activity. |
| + Anti-TLR4 Blocking Ab | Partial Reduction (e.g., 40-70%) | Partial Reduction | Confirms TLR4 mediates a significant fraction of signaling. |
| + Anti-RAGE Blocking Ab | Partial Reduction (e.g., 30-60%) | Partial Reduction | Confirms RAGE mediates a significant fraction of signaling. |
| + Dual TLR4/RAGE Block | Near-Complete Abrogation (>90%) | Near-Complete Abrogation | Confirms signaling is primarily synergistic via both receptors. |
| + Isotype Control Ab | No Change | No Change | Confirms specific antibody effect. |
| + TAK-242 (TAK-242) | Complete Abrogation of TLR4-mediated signal | Complete Abrogation of TLR4-mediated signal | Pharmacological confirmation of TLR4 pathway role. |
Conclusion: Meticulous antibody selection, coupled with rigorously controlled competitive binding and functional blocking assays, is non-negotiable for defining the discrete roles of TLR4 and RAGE in HMGB1-driven sterile inflammation. The protocols and frameworks outlined here provide a roadmap for generating robust, interpretable data to advance therapeutic development in this complex signaling axis.
Within the research on sterile inflammatory processes—such as ischemia-reperfusion injury, trauma, and non-infectious organ failure—the alarmin High Mobility Group Box 1 (HMGB1) is a critical mediator. Its biological activity is context-dependent, determined by post-translational modifications (redox state, acetylation) and its interaction with key receptors, primarily Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End products (RAGE). Achieving data reproducibility in HMGB1 studies is fundamentally challenged by the source and preparation of the HMGB1 protein used. This guide provides a technical framework for standardizing HMGB1 preparation, comparing recombinant and endogenous sources, to ensure reliable and interpretable results in HMGB1-TLR4-RAGE signaling research.
The choice between recombinant and endogenous HMGB1 profoundly impacts experimental outcomes. The table below summarizes key characteristics.
Table 1: Comparative Analysis of Recombinant vs. Endogenous HMGB1 Preparations
| Characteristic | Recombinant HMGB1 (E.g., E. coli, mammalian) | Endogenous HMGB1 (From cell culture supernatants or tissue) |
|---|---|---|
| Source | Heterologous expression systems (E. coli, yeast, mammalian cells). | Secreted/released from primary cells or cell lines (e.g., LPS-stimulated macrophages). |
| Purity & Homogeneity | High (≥95%). Homogeneous amino acid sequence. | Variable. Contaminated with other secreted alarmins (e.g., S100s, HSPs). |
| Post-Translational Modifications (PTMs) | Lacks mammalian PTMs unless produced in mammalian systems. Often fully reduced. | Contains native PTMs (acetylation, phosphorylation). Redox states (fully reduced, disulfide, oxidized) are mixed and biologically relevant. |
| Reproducibility | High between batches from same vendor/protocol. | Low. Highly dependent on cell type, stimulus, and purification method. |
| Biological Activity | Can be potent but may not reflect native activity due to lack of PTMs. | More physiologically relevant but activity is heterogeneous. |
| Key Advantages | Defined concentration, high purity, scalable, suitable for structural studies. | Authentic redox isoforms and PTM patterns, relevant for in vivo modeling. |
| Primary Disadvantage | Non-physiological redox state (often fully reduced) may skew receptor usage. | Uncontrolled co-contaminants can confound signaling attribution. |
| Cost & Time | Moderate cost, quick acquisition. | High time investment, requires validation, variable cost. |
Standardized Experimental Protocols
Protocol for Active, Disulfide HMGB1 Production from Recombinant Protein
Adapted from Venereau et al., 2012 & Antoine et al., 2014.
Objective: To convert commercially available, fully reduced recombinant HMGB1 into the inflammatory, disulfide isoform (C23-C45 disulfide bond) that signals via TLR4.
- Material: Lyophilized, E. coli-derived, endotoxin-free recombinant human HMGB1.
- Reduction: Dissolve HMGB1 (1 mg/mL) in degassed 50 mM Tris-HCl, 20 mM NaCl, pH 8.0, with 5 mM DTT. Incubate 2 hours at 37°C under argon.
- Desalting: Pass the solution through a Zeba Spin Desalting Column (7K MWCO) pre-equilibrated with degassed buffer (without DTT) to remove reducing agent.
- Oxidation/Folding: Immediately dilute the reduced protein into a 100-fold molar excess of oxidized glutathione (GSSG) in degassed buffer. Final GSSG concentration: ~2 mM. Incubate for 16-24 hours at 4°C under argon.
- Verification: Confirm disulfide bond formation by non-reducing SDS-PAGE (slight gel shift) and mass spectrometry. Validate biological activity in a TLR4-dependent cell assay (e.g., NF-κB luciferase in HEK-Blue TLR4 cells).
Protocol for Endogenous HMGB1 Isolation from Cell Culture
Adapted from Lotze & Tracey, 2005 and recent methodologies.
Objective: To purify HMGB1 released from stimulated macrophages, preserving its native redox isoforms.
- Cell Stimulation: Culture RAW 264.7 or primary murine macrophages. Stimulate with LPS (100 ng/mL) for 12-16 hours. Optional: Add ethyl pyruvate (10-20 mM) to inhibit release for a negative control.
- Supernatant Collection: Centrifuge culture media (300 x g, 10 min) to remove cells. Add protease inhibitors (e.g., PMSF, aprotinin) immediately.
- Concentration & Dialysis: Concentrate supernatant 20-50 fold using a 10 kDa MWCO centrifugal concentrator. Dialyze against 20 mM Tris-HCl, 50 mM NaCl, pH 7.4.
- Heparin-Sepharose Affinity Chromatography: Load dialyzed supernatant onto a heparin-Sepharose column. Wash with 10 column volumes of dialysis buffer. Elute HMGB1 with a linear NaCl gradient (50 mM to 2 M) in Tris buffer. HMGB1 typically elutes between 0.5-1 M NaCl.
- Validation: Analyze fractions by SDS-PAGE and Western Blot (anti-HMGB1). Test for endotoxin contamination (<0.1 EU/µg). Characterize redox state via HPLC or non-reducing diagonal gel electrophoresis.
Visualization of Key Concepts
HMGB1 Isoforms and Receptor Signaling Pathways
Diagram Title: HMGB1 Source Determines Isoform and Receptor Pathway
Experimental Workflow for HMGB1 Standardization
Diagram Title: HMGB1 Standardization and Validation Workflow
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for HMGB1 Reproducibility Research
| Reagent / Material | Function & Rationale | Example Vendor / Catalog Consideration |
|---|---|---|
| Endotoxin-Free Recombinant HMGB1 | Base material for generating defined redox isoforms. Must be <0.1 EU/µg to avoid TLR4 confounding by LPS. | R&D Systems (human, mouse), HMGBiotech (redox mutants) |
| HEK-Blue TLR4 or TLR2 Cells | Reporter cell lines for specific, quantifiable assessment of HMGB1-TLR4 activity, segregating it from RAGE signaling. | InvivoGen |
| Heparin-Sepharose 6 Fast Flow | Affinity resin for purifying endogenous HMGB1 based on its high heparin-binding affinity. | Cytiva |
| Zeba Spin Desalting Columns | For rapid buffer exchange to remove DTT/GSSG during redox refolding of recombinant protein, preventing scrambling. | Thermo Fisher Scientific |
| Oxidized (GSSG) & Reduced (GSH) Glutathione | Critical for in vitro redox refolding to produce the disulfide HMGB1 isoform. | Sigma-Aldrich |
| LAL Endotoxin Assay Kit | Essential for quantifying endotoxin contamination in all HMGB1 prep, a mandatory validation step. | Lonza, Charles River Laboratories |
| Anti-HMGB1 Antibodies (Non-Redox Specific) | For detection and quantification via Western Blot or ELISA across all isoforms. | Abcam, Cell Signaling Technology |
| RAGE Inhibitor (e.g., FPS-ZM1) | Pharmacological tool to dissect RAGE-specific effects from TLR4 signaling in bioassays. | EMD Millipore |
| TLR4 Inhibitor (e.g., TAK-242) | Pharmacological tool to inhibit TLR4 signaling and confirm HMGB1 activity is TLR4-dependent. | InvivoGen, MedChemExpress |
| Protease Inhibitor Cocktail | Prevents degradation of endogenous HMGB1 during purification from cell supernatants or tissue. | Roche, Thermo Fisher Scientific |
Sterile inflammation, driven by damage-associated molecular patterns (DAMPs) like HMGB1, is a critical pathological process in ischemia-reperfusion injury, trauma, and non-infectious degenerative diseases. The HMGB1-TLR4-RAGE signaling axis is a central focus of this research. Selecting the appropriate in vivo model system is paramount for generating translatable, mechanistic insights. This guide provides a technical framework for model selection, grounded in the specific requirements of HMGB1 pathway research.
Core Signaling Context: HMGB1, TLR4, and RAGE
High Mobility Group Box 1 (HMGB1) is a prototypical DAMP released from necrotic or stressed cells. It signals primarily through Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End-products (RAGE), leading to NF-κB activation, cytokine production, and sustained inflammation. The choice of in vivo model must allow for dissection of this specific axis.
HMGB1 Signaling via TLR4 and RAGE
Comparative Analysis ofIn VivoModel Systems
The following table summarizes key in vivo systems for studying sterile inflammation, with metrics relevant to HMGB1-TLR4-RAGE research.
Table 1: In Vivo Model Systems for Sterile Inflammation Research
| Model System | Key Sterile Injury Inductions | Relevance to HMGB1/TLR4/RAGE | Throughput | Genetic Tractability | Cost & Infrastructure | Key Limitations |
|---|---|---|---|---|---|---|
| Mouse (Mus musculus) | Hepatic I/R, Myocardial I/R, Trauma, Bilateral Nephrectomy | Excellent: Wide array of KO (TLR4⁻/⁻, RAGE⁻/⁻), conditional, reporter strains available. | Moderate to High | Very High | Moderate | Immune system differences from humans. |
| Rat (Rattus norvegicus) | Similar to mouse; favored for organ I/R, hemorrhagic shock. | Good: Pharmacologic inhibitors well-established. KO strains less available than mouse. | Moderate | Low to Moderate | Moderate | Less genetic toolbox than mouse. |
| Zebrafish (Danio rerio) | Tail fin amputation, cryoinjury, chemical injury. | Emerging: HMGB1 orthologs exist; TLR4 function debated; real-time imaging of inflammation is key strength. | Very High | High (transgenics, morpholinos) | Low | Differing immune architecture; not mammalian. |
| Porcine (Sus scrofa) | Myocardial I/R, Trauma/Surgery models. | Good: Physiologically similar to human; suitable for pre-clinical therapeutic testing. | Low | Very Low | Very High | Limited reagents, expensive, low throughput. |
Table 2: Quantitative Metrics for Common Sterile Injury Models in Mice
| Injury Model | Onset of HMGB1 Release (Post-Injury) | Peak Inflammatory Cytokine Response | Key Readouts | Model Fidelity to Human Condition |
|---|---|---|---|---|
| Hepatic Ischemia/Reperfusion | 1-3 hours | 6-24 hours | Serum ALT/AST, liver histology, neutrophil infiltration, cytokines. | High for liver transplant, shock. |
| Myocardial I/R (LAD Ligation) | 1-2 hours | 12-48 hours | Infarct size (TTC staining), echocardiography, troponin, cardiac cytokines. | High for acute MI. |
| Traumatic Brain Injury (Controlled CCI) | Immediate - 6 hours | 24-72 hours | Neurological scores, brain water content, histology, CNS cytokines. | Moderate to High. |
| Sterile Chemical Injury (Acetaminophen) | 6-12 hours | 12-48 hours | Serum ALT, liver necrosis area, HMGB1 localization (nucleus to cytoplasm). | High for drug-induced liver injury. |
Detailed Experimental Protocols
Protocol 1: Mouse Model of Hepatic Ischemia/Reperfusion (Partial, 70%)
Purpose: To induce sterile inflammatory injury in the liver, a key organ for HMGB1 release studies.
Materials:
- Anesthetized C57BL/6 mouse (or relevant transgenic).
- Warm saline pad.
- Sterile surgical tools (micro-scissors, atraumatic clamps).
- 6-0 silk suture.
- Isoflurane anesthesia system.
Method:
- Anesthetize mouse and place supine on heating pad.
- Perform midline laparotomy. Gently exteriorize the liver.
- Identify the portal triad to the left and median lobes. Occlude the blood flow using a non-traumatic microvascular clamp for 60 minutes (ischemic phase). Ensure occlusion is complete.
- Cover exposed organs with sterile saline-moistened gauze. Maintain body temperature at 37°C.
- After 60 minutes, remove the clamp to initiate reperfusion. Confirm return of blood flow.
- Suture the abdominal wall and skin in two layers.
- At designated reperfusion timepoints (e.g., 2h, 6h, 24h), collect serum and liver tissue.
- Key Analyses: Serum ALT/AST (necrosis), liver histology (H&E), immunohistochemistry for HMGB1 (translocation), ELISA for cytokines (TNF-α, IL-6), and Western blot for phospho-NF-κB p65 in nuclear extracts.
Protocol 2: Assessment of HMGB1 Release and Pathway Activation
Purpose: To quantify HMGB1 release and downstream signaling in tissue or serum samples.
Materials:
- Tissue homogenizer.
- ELISA kits: HMGB1, TNF-α, IL-6.
- Antibodies for Western Blot: anti-HMGB1, anti-phospho-NF-κB p65, anti-TLR4, anti-RAGE, appropriate HRP-conjugated secondaries.
- Nuclear extraction kit.
Method (Serum/Tissue HMGB1 ELISA):
- Collect blood, allow to clot, centrifuge for serum. For tissue, homogenize in protease-inhibitor containing PBS.
- Follow commercial HMGB1 ELISA protocol (e.g., IBL International or Shino-Test). Typically involves: coating capture Ab, blocking, adding samples and standards, adding detection Ab, adding substrate, and reading absorbance.
- Critical Note: Use EDTA-plasma or serum processed quickly, as HMGB1 can bind to fibrinogen.
Method (Pathway Analysis via Western Blot):
- Homogenize tissue in RIPA buffer with protease/phosphatase inhibitors.
- Separate proteins by SDS-PAGE, transfer to PVDF membrane.
- Block with 5% BSA/TBST. Incubate with primary antibody overnight at 4°C.
- Incubate with HRP-secondary antibody, develop with ECL.
- For Nuclear NF-κB: Use a commercial nuclear extraction kit to isolate nuclear fractions before Western blotting for phospho-p65.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for HMGB1-TLR4-RAGE In Vivo Research
| Reagent | Function & Application | Example Product/Catalog # (for reference) |
|---|---|---|
| Recombinant HMGB1 | Used as a positive control or to directly induce signaling in challenge experiments. | HMGB1 protein, endotoxin-free (e.g., R&D Systems, 1690-HMB). |
| Anti-HMGB1 Neutralizing Antibody | To therapeutically block HMGB1 activity in vivo; validates HMGB1's role. | Anti-HMGB1 mAb (e.g., BioLegend, 651402). |
| TLR4 Inhibitor (TAK-242/Resatorvid) | Small molecule inhibitor of TLR4 signaling; used to dissect TLR4-specific effects. | TAK-242 (Cayman Chemical, 19910). |
| RAGE Antagonist (FPS-ZM1) | High-affinity RAGE-specific inhibitor; used to block RAGE-mediated signaling. | FPS-ZM1 (MilliporeSigma, 553030). |
| TLR4 KO & RAGE KO Mice | Genetic models to definitively assign signaling pathways. | Available from Jackson Laboratory (e.g., B6.B10ScN-Tlr4 |
| HMGB1 Reporter Mice | Allows spatial and temporal tracking of HMGB1 expression in vivo. | Not widely commercial; often generated in-house. |
| Phospho-NF-κB p65 (Ser536) Antibody | Key readout for downstream pathway activation via Western blot or IHC. | Cell Signaling Technology, #3033. |
| Multiplex Cytokine Panel | To quantify the inflammatory cascade resulting from HMGB1 signaling. | Mouse Premixed Multi-Analyte Kit (e.g., R&D Systems, LXSAHM). |
Model Selection Workflow and Decision Logic
Decision Logic for In Vivo Model Selection
The optimal in vivo system for sterile inflammation research hinges on the specific question within the HMGB1-TLR4-RAGE axis. Mice remain the gold standard for mechanistic, genetic studies. Zebrafish offer unparalleled visual access to inflammatory dynamics. Large animals provide critical translational bridges. Aligning the model's strengths—genetic tractability, physiological relevance, throughput, and cost—with the experimental goals is essential for generating robust, meaningful data that advances our understanding of sterile inflammatory diseases.
Therapeutic Targeting and Validation: Comparing Strategies to Disrupt Sterile Inflammation
High Mobility Group Box 1 (HMGB1) is a prototypic Damage-Associated Molecular Pattern (DAMP) molecule that plays a pivotal role in sterile inflammation, a driver of pathology in conditions like ischemia-reperfusion injury, sepsis, and autoimmune diseases. Its signaling axis, primarily through Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End products (RAGE), orchestrates a pro-inflammatory cytokine cascade. This whitepaper details the three principal classes of direct HMGB1 inhibitors—neutralizing antibodies, peptides, and heparin derivatives—framed within the ongoing thesis that targeting extracellular HMGB1’s interaction with TLR4 and RAGE is a validated therapeutic strategy for sterile inflammatory diseases.
Inhibition Mechanisms and Quantitative Efficacy
The following table summarizes the core mechanisms and quantitative efficacy data for the three major classes of HMGB1 inhibitors.
Table 1: Comparative Analysis of Direct HMGB1 Inhibitor Classes
| Inhibitor Class | Prototype Example | Primary Target/MoA | Reported In Vivo Efficacy (Key Models) | Development Stage |
|---|---|---|---|---|
| Neutralizing Antibodies | Anti-HMGB1 mAb (2G7) | Binds to HMGB1, blocks interaction with TLR4/RAGE. | ~60% reduction in serum TNF-α in murine sepsis; ~40% reduction in infarct size in murine MI/R. | Preclinical / Phase I-II (for sepsis, ARDS). |
| Peptides | Box A (HMG-box A domain) | Acts as a competitive antagonist for full-length HMGB1 at RAGE. | ~50% reduction in neutrophil infiltration in murine hepatic I/R; significant improvement in survival in murine sepsis. | Preclinical. |
| Heparin Derivatives | Non-anticoagulant heparin (SST0001) | Binds to HMGB1's heparin-binding domain, preventing receptor engagement. | ~70% reduction in HMGB1-mediated vascular leakage; ~55% reduction in arthritis score in murine CIA model. | Preclinical. |
Experimental Protocols for Key Assays
Protocol 1: ELISA-Based HMGB1 Neutralization Assay
- Objective: To quantify the ability of an inhibitor (antibody, peptide, or heparin) to block HMGB1 binding to its receptors.
- Materials: Recombinant HMGB1, soluble RAGE or TLR4-Fc chimera, inhibitor samples, 96-well plate coated with HMGB1, detection antibody (anti-RAGE or anti-Fc-HRP), TMB substrate, plate reader.
- Procedure:
- Coat plate with HMGB1 (1 µg/mL, 100 µL/well) overnight at 4°C.
- Block with 1% BSA/PBS for 2 hours.
- Pre-incubate a fixed concentration of HMGB1 (10 nM) with serially diluted inhibitors for 1 hour at 37°C.
- Add the HMGB1-inhibitor mix to the coated wells. Include HMGB1-only (max signal) and no-HMGB1 (blank) controls.
- Incubate for 2 hours, wash.
- Add detection antibody (1:2000), incubate 1 hour, wash.
- Develop with TMB, stop with H₂SO₄, read absorbance at 450 nm.
- Calculate % inhibition relative to HMGB1-only control.
Protocol 2: In Vitro Barrier Function Assay (for Heparin Derivatives)
- Objective: To assess inhibition of HMGB1-induced endothelial hyperpermeability.
- Materials: Human Umbilical Vein Endothelial Cells (HUVECs), Transwell inserts (3.0 µm pore), FITC-dextran (40 kDa), recombinant HMGB1, test heparin derivative, fluorometer.
- Procedure:
- Seed HUVECs on Transwell inserts and culture to form a confluent monolayer (typically 2-3 days).
- Pre-treat the basolateral compartment with inhibitor (e.g., 100 µg/mL SST0001) for 30 min.
- Stimulate with HMGB1 (10 ng/mL) added to the basolateral compartment for 16-24 hours.
- Add FITC-dextran to the apical compartment.
- Measure fluorescence in the basolateral compartment at 60-minute intervals.
- Calculate permeability coefficient (Papp) and % inhibition relative to HMGB1-treated controls.
Signaling Pathways and Experimental Workflows
HMGB1 Signaling Pathways and Inhibitor Mechanisms
HMGB1 Inhibitor Screening Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for HMGB1-TLR4/RAGE Research
| Reagent / Material | Function & Application | Example Vendor/Clone |
|---|---|---|
| Recombinant HMGB1 | Full-length or domain-specific (Box A, Box B). Used as stimulus in in vitro and in vivo models. | R&D Systems, Sigma-Aldrich. |
| Anti-HMGB1 Neutralizing mAb | For immunoneutralization, ELISA detection, and Western blotting. Clone 2G7 is widely cited. | BioLegend (Clone 2G7), Abcam. |
| Soluble RAGE (sRAGE) Fc | Decoy receptor; used as positive control for inhibition or to detect HMGB1 binding in ELISA. | Sino Biological, R&D Systems. |
| TLR4/MD2 Reporter Cell Line | Engineered cells (e.g., HEK293) with TLR4/MD2 and inducible luciferase. For functional HMGB1 signaling assays. | InvivoGen (HEK-Blue hTLR4). |
| Non-Anticoagulant Heparin (NAH) | Heparin derivative lacking anti-FXa activity. Key tool for dissecting HMGB1-heparin domain interactions. | Sigma-Aldrich (Heparinase-treated), SST0001 (research grade). |
| HMGB1 ELISA Kit | Quantifies total or disulfide HMGB1 in serum, plasma, or cell culture supernatants. | IBL International, Tecan. |
This whitepaper provides a technical comparison of two principal therapeutic strategies for modulating the HMGB1-mediated signaling axis in sterile inflammation. The damage-associated molecular pattern (DAMP) HMGB1 propagates inflammatory responses primarily via two receptors: Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End products (RAGE). Within the broader thesis of HMGB1/TLR4/RAGE signaling in sterile inflammation (e.g., ischemia-reperfusion injury, non-infectious sepsis, autoimmune diseases), targeted antagonism of these receptors presents distinct mechanistic pathways, efficacy profiles, and challenges. This guide details the current landscape of TLR4 inhibitors and RAGE antagonists, supported by experimental data and methodologies.
Signaling Pathways: HMGB1, TLR4, and RAGE
HMGB1 signaling is context-dependent. TLR4 activation, often requiring co-factors like MD-2, primarily drives pro-inflammatory cytokine production (e.g., TNF-α, IL-6) via MyD88/NF-κB and TRIF/IRF3 pathways. RAGE engagement, a multi-ligand receptor, activates sustained NF-κB and MAPK signaling, contributing to oxidative stress and cellular dysfunction. Cross-talk between these pathways creates a positive feedback loop amplifying inflammation.
Diagram: HMGB1 Signaling Axis in Sterile Inflammation
TLR4 Inhibitors: Mechanisms and Profiles
TLR4 inhibitors target various points in the receptor complex. Small molecules (e.g., TAK-242/Resatorvid, Eritoran) bind MD-2 or TLR4 intracellularly to disrupt signaling. Antibodies and peptide inhibitors block ligand binding or receptor dimerization.
Table 1: Representative TLR4 Inhibitors - Efficacy & Side-Effects
| Compound (Class) | Primary Target | Key Efficacy Findings (Model) | Major Reported Side-Effects / Limitations |
|---|---|---|---|
| TAK-242 (Resatorvid) | Intracellular TLR4 domain | Reduced mortality & cytokines in murine sepsis; improved cardiac function in MI models. | Clinical trials in sepsis failed (hepatotoxicity, lack of efficacy in late-stage patients). |
| Eritoran (E5564) | MD-2 co-receptor | Antagonized LPS & HMGB1; effective in early preclinical sepsis & sterile liver injury. | No mortality benefit in Phase III sepsis trials (possibly wrong patient population/timing). |
| NI-0101 (mAb) | TLR4 dimerization | Blocked LPS & HMGB1 signaling in human cell assays; reduced inflammation in RA models. | Clinical development halted (lack of efficacy in rheumatoid arthritis trial). |
| RSLA (Peptide) | TLR4 extracellular | Attenuated neuroinflammation & white matter injury in rodent stroke models. | Limited bioavailability; primarily preclinical. |
RAGE Antagonists: Mechanisms and Profiles
RAGE antagonists include soluble RAGE (sRAGE, a decoy receptor), small-molecule antagonists (e.g., Azeliragon, FPS-ZM1), and anti-RAGE antibodies. They aim to block ligand binding, inhibiting downstream pro-inflammatory and pro-migratory signals.
Table 2: Representative RAGE Antagonists - Efficacy & Side-Effects
| Compound (Class) | Primary Target | Key Efficacy Findings (Model) | Major Reported Side-Effects / Limitations |
|---|---|---|---|
| sRAGE (Decoy) | Extracellular ligand binding | Reduced vascular inflammation, atherosclerosis, & tumor growth in murine models. | Pharmacokinetic challenges (short half-life, high production cost); not orally available. |
| Azeliragon (TTP488) | Transmembrane RAGE | Phase 3 for Alzheimer's (mild); showed reduced cognitive decline in subpopulation. | Increased adverse events (infections, CV events) in broader trial; narrow therapeutic window. |
| FPS-ZM1 (Small Molecule) | V domain of RAGE | High efficacy in reducing Aβ plaque & neuroinflammation in AD mouse models. | Preclinical stage; long-term safety profile in humans unknown. |
| Anti-RAGE mAb | Extracellular domain | Attenuated diabetic atherosclerosis & restenosis in animal models. | Potential immunogenicity; limited clinical data available. |
Comparative Experimental Protocols
Protocol 1: Assessing Inhibitor Efficacy on HMGB1-Induced NF-κB Activation
- Objective: Quantify and compare the inhibitory potency of TLR4 vs. RAGE antagonists on HMGB1-driven signaling.
- Cell Line: Human THP-1 monocytes (differentiated with PMA) or primary macrophages.
- Reagents: Recombinant HMGB1 (100 ng/mL), TAK-242 (TLR4 inhibitor, 1µM), FPS-ZM1 (RAGE antagonist, 1µM), NF-κB luciferase reporter plasmid, transfection reagent, luciferase assay kit.
- Method:
- Transfect cells with NF-κB luciferase reporter construct.
- Pre-treat cells with antagonist or vehicle control for 1 hour.
- Stimulate with recombinant HMGB1 for 6 hours.
- Lyse cells and measure luciferase activity.
- Data Analysis: Normalize to protein content. Express data as % inhibition relative to HMGB1-stimulated, untreated control.
Protocol 2: Cytokine Profiling in a Sterile Inflammation Model (e.g., Liver I/R)
- Objective: Compare in vivo efficacy of leading candidates from each class.
- Animal Model: Murine hepatic ischemia-reperfusion model (60 min ischemia, 6-24h reperfusion).
- Dosing: Antagonist (e.g., Eritoran, 3 mg/kg; or sRAGE, 100 µg/mouse) administered intravenously prior to reperfusion.
- Sample Collection: Serum and liver tissue at sacrifice.
- Analysis:
- ELISA: Quantify serum TNF-α, IL-6, HMGB1.
- qPCR: Liver tissue mRNA for Cxcl1, Cxcl2, Icam1.
- Histology: H&E staining for necrotic area quantification.
- Comparison Metrics: Percent reduction in serum cytokines, injury markers, and histologic damage vs. vehicle-treated I/R controls.
Diagram: In Vivo Efficacy Testing Workflow
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for HMGB1-TLR4/RAGE Research
| Reagent | Function/Application | Example Product (Vendor) |
|---|---|---|
| Recombinant HMGB1 | Primary DAMP for in vitro and in vivo stimulation. Must use endotoxin-free (<0.1 EU/µg) prep. | Human HMGB1, endotoxin-free (R&D Systems, Sigma). |
| TLR4/MD-2 Inhibitors | Tool compounds for mechanistic studies; positive controls. | TAK-242 (InvivoGen), Eritoran (Cayman Chemical). |
| RAGE Antagonists | Tool compounds to dissect RAGE-specific signaling. | FPS-ZM1 (Tocris), Recombinant sRAGE (R&D Systems). |
| Phospho-Specific Antibodies | Detect activation of downstream pathways (NF-κB p65, MAPKs). | Anti-phospho-NF-κB p65 (S536) (Cell Signaling). |
| sRAGE ELISA Kit | Measure circulating sRAGE as a potential biomarker or pharmacokinetic marker. | Human sRAGE Quantikine ELISA (R&D Systems). |
| NF-κB Reporter Cell Line | Stable reporter for high-throughput screening of inhibitors. | THP-1-Blue NF-κB cells (InvivoGen). |
| Anti-HMGB1 Neutralizing Ab | Control for HMGB1-specific effects. | Monoclonal anti-HMGB1 (BioLegend). |
| LPS-RS (TLR4 antagonist) | Negative control for LPS contamination in HMGB1 preps. | LPS from R. sphaeroides (InvivoGen). |
Direct comparison reveals a trade-off: TLR4 inhibitors offer potent, rapid suppression of acute cytokine storms but have faced clinical challenges in heterogeneous conditions like sepsis. RAGE antagonists may provide broader modulation of chronic inflammation and cellular dysfunction but can have on-target side effects due to RAGE's role in homeostasis. The future of therapeutics in sterile inflammation may lie in combination strategies—simultaneously targeting HMGB1 release, TLR4 signaling, and RAGE activation—or in patient stratification based on dominant signaling pathways. Rigorous preclinical models that accurately reflect human disease pathophysiology are essential for advancing the next generation of antagonists in this field.
The high-mobility group box 1 (HMGB1) protein is a critical damage-associated molecular pattern (DAMP) driving sterile inflammation via its receptors, primarily Toll-like receptor 4 (TLR4) and the receptor for advanced glycation end products (RAGE). This signaling axis is a pivotal therapeutic target in conditions like sepsis, ischemia-reperfusion injury, and autoimmune diseases. Upon HMGB1 binding, TLR4 initiates two distinct downstream pathways: the Myeloid Differentiation Primary Response 88 (MyD88)-dependent pathway and the TIR-domain-containing adapter-inducing interferon-β (TRIF)-dependent pathway. Both converge on Mitogen-Activated Protein Kinase (MAPK) cascades (ERK, p38, JNK) and NF-κB, leading to pro-inflammatory cytokine production. Effective blockade of these downstream signals offers a strategic approach to modulate pathological inflammation while potentially preserving essential immune function.
Signaling Pathways: Core Targets for Inhibition
Diagram of HMGB1/TLR4/RAGE Downstream Signaling & Inhibition Points
Diagram Title: HMGB1 TLR4 RAGE Signaling with Inhibitor Points
Quantitative Data on Key Inhibitors
Table 1: Pharmacological Inhibitors of MyD88, TRIF, and MAPKs
| Target | Exemplary Inhibitor | Chemical Nature | IC50 / Kd | Key Cellular Assay Readout | Known Off-Target Effects |
|---|---|---|---|---|---|
| MyD88 | ST2825 (Myd88 homodimerization inhibitor) | Peptidomimetic | IC50: ~10 µM (dimerization) | ↓ LPS-induced IL-6, TNF-α in macrophages | IRAK1/4 inhibition at high conc. |
| MyD88 | TJ-M2010-5 | Small molecule | IC50: 4.7 µM (HEK-Blue hTLR4 assay) | ↓ NF-κB luciferase reporter activity | Limited data |
| TRIF | E6446 (also inhibits TLR9) | Quinoline derivative | IC50: 2-10 µM (TLR4-TRIF in HEK cells) | ↓ Poly(I:C)-induced IFN-β; ↓ LPS late-phase IRF3 activation | TLR7/9 inhibition |
| TAK1 | 5Z-7-Oxozeaenol | Resorcylic acid lactone | Kd: ~3.3 nM (irreversible) | ↓ Phospho-p38, phospho-JNK; ↓ IL-1β-induced IL-8 | Covalent binder; potential general kinase reactivity |
| ERK1/2 | SCH772984 | Pyrimidine derivative | IC50: 4 nM (ERK1), 1 nM (ERK2) | ↓ Phospho-RSK1 (S380); cell cycle arrest | p38 MAPK at >1 µM |
| p38α/β | SB203580 | Pyridinyl imidazole | IC50: ~50 nM (p38α/β) | ↓ LPS-induced TNF-α; ↓ Phospho-ATF2 | Inhibits CK1δ/ε, GSK-3β, JAK2 at high µM |
| JNK | SP600125 | Anthrapyrazolone | IC50: ~40 nM (JNK1), ~90 nM (JNK2) | ↓ c-Jun phosphorylation (Ser63); ↓ AP-1 activity | Inhibits AMPK, CK1δ, PIM1, others |
Table 2: Functional Outcomes in HMGB1-Induced Sterile Inflammation Models
| Inhibitor (Target) | In Vitro Model | Key Effect (Concentration) | In Vivo Model (e.g., Sepsis, I/R) | Outcome (Dose, Route) |
|---|---|---|---|---|
| ST2825 (MyD88) | RAW264.7 macrophages + HMGB1 | ↓ TNF-α by 75% (20 µM) | Cecal ligation & puncture (CLP) mouse | ↑ Survival 40% → 70% (5 mg/kg, i.p.) |
| E6446 (TRIF) | BMDMs + HMGB1/LPS | ↓ Late-phase IFN-β by 60% (10 µM) | Hepatic I/R injury mouse | ↓ Serum ALT by 50%, ↓ Necrosis (10 mg/kg, i.v.) |
| 5Z-7-Oxozeaenol (TAK1) | HMEC-1 endothelial cells + HMGB1 | ↓ IL-8 secretion by 85% (100 nM) | Murine model of sterile arthritis | ↓ Joint swelling by 60% (1 mg/kg, i.p.) |
| SB203580 (p38) | PBMCs from septic patients | ↓ IL-1β production by 65% (5 µM) | LPS-induced endotoxemia mouse | ↓ Plasma IL-6, attenuated shock (30 mg/kg, i.p.) |
Experimental Protocols for Evaluation
Protocol: Evaluating MyD88 Inhibitors in HMGB1-Stimulated Macrophages
Objective: To assess the efficacy of MyD88 dimerization inhibitors on pro-inflammatory cytokine production.
Materials:
- Primary mouse Bone Marrow-Derived Macrophages (BMDMs) or RAW264.7 cells.
- Recombinant HMGB1 (≥95% endotoxin-free).
- Inhibitors: ST2825 (Tocris), TJ-M2010-5 (MedChemExpress), vehicle control (DMSO).
- LPS-RS (TLR4 antagonist) as a control for specificity.
- ELISA kits for mouse TNF-α, IL-6.
- Cell viability assay (MTT or CellTiter-Glo).
- Western blot reagents: antibodies for phospho-IκBα, total IκBα, β-actin.
Procedure:
- Cell Preparation & Pretreatment: Seed cells in 24-well plates (2.5x10^5 cells/well). After adherence, pre-treat cells with varying concentrations of inhibitor (e.g., 1, 5, 20 µM) or vehicle (DMSO, final conc. ≤0.1%) for 1 hour.
- Stimulation: Add recombinant HMGB1 (final conc. 100 ng/mL) or ultrapure LPS (10 ng/mL, positive control) for 6 hours (mRNA) or 16 hours (secreted protein).
- Harvest: Collect supernatant for ELISA. Lyse cells in RIPA buffer for Western blot analysis of NF-κB pathway activation (degradation of IκBα).
- Controls: Include wells with (a) medium only (basal), (b) HMGB1 + vehicle, (c) HMGB1 + LPS-RS (10 µg/mL), (d) inhibitor only (toxicity control).
- Analysis: Normalize cytokine concentrations to cell viability. Express data as % inhibition relative to HMGB1+vehicle control.
Protocol: MAPK Inhibition Profiling via Phospho-Kinase Array
Objective: To simultaneously profile the effect of a candidate inhibitor on multiple kinase phosphorylation nodes downstream of HMGB1/TLR4.
Materials:
- Proteome Profiler Mouse Phospho-Kinase Array Kit (ARY003B, R&D Systems).
- Cell lysates from HMGB1-stimulated macrophages with/without inhibitor.
- Detection reagents from kit.
- Chemiluminescence imager.
Procedure:
- Stimulation & Lysis: Treat cells (e.g., J774A.1 macrophage line) with inhibitor or vehicle for 1h, then stimulate with HMGB1 (100 ng/mL) for 15-30 minutes. Lyse cells using the kit's lysis buffer.
- Array Incubation: Mix lysates (300-500 µg protein) with detection antibody cocktail. Apply to the array membrane. Incubate overnight at 4°C on a rocking platform.
- Wash & Detection: Wash membranes thoroughly. Incubate with Streptavidin-HRP for 30 min. Develop using chemiluminescent substrate.
- Data Quantification: Spot density is quantified using ImageJ software. Normalize phospho-signals to reference spots on the membrane. Compare inhibitor-treated vs. vehicle-treated, HMGB1-stimulated samples to identify specific pathway blockade (e.g., p-p38, p-JNK, p-ERK).
Research Reagent Solutions Toolkit
Table 3: Essential Reagents for Downstream Signal Blockade Studies
| Reagent / Material | Supplier Examples | Function in Experiments |
|---|---|---|
| Recombinant HMGB1 (Endotoxin-free) | HMGBiotech, R&D Systems | The primary DAMP for specific stimulation of TLR4/RAGE pathways in sterile inflammation models. |
| TLR4 Inhibitors (LPS-RS, TAK-242) | InvivoGen, Sigma | Critical controls to confirm TLR4-dependent effects versus RAGE or other receptors. |
| MyD88 Inhibitor (ST2825) | Tocris, MedChemExpress | Tool compound to block the MyD88-dependent branch of TLR4 signaling. |
| TRIF Inhibitor (E6446) | Sigma, InvivoGen | Tool compound to block the TRIF-dependent branch, assessing IRF3/IFN-β responses. |
| MAPK Inhibitor Set (SB203580, SP600125, SCH772984) | Selleckchem, Cayman Chemical | Selective inhibitors for p38, JNK, and ERK to dissect contributions of specific MAPK pathways. |
| Phospho-Specific Antibodies (p-p38, p-JNK, p-ERK, p-IκBα) | Cell Signaling Technology | Key for Western blot validation of inhibitor efficacy on target phosphorylation. |
| Phospho-Kinase Array Kit | R&D Systems | Multiplexed screening tool to evaluate inhibitor effects across multiple signaling nodes simultaneously. |
| NF-κB/AP-1 Luciferase Reporter Cell Line | InvivoGen (HEK-Blue hTLR4) | High-throughput assay for functional readout of MyD88/NF-κB and TRIF/IRF pathway activity. |
| Cytokine ELISA Kits (Mouse/Rat TNF-α, IL-6, IL-1β, IFN-β) | BioLegend, R&D Systems | Quantification of the ultimate functional output of the signaling cascade. |
The transition from promising preclinical data to successful clinical outcomes remains a formidable challenge in drug development, particularly in complex fields like sterile inflammation. The HMGB1-TLR4-RAGE signaling axis serves as a paradigm for this challenge. High Mobility Group Box 1 (HMGB1), acting as a Damage-Associated Molecular Pattern (DAMP), propagates inflammation via receptors like Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End products (RAGE). Despite robust evidence linking this pathway to pathologies like sepsis, ischemia-reperfusion injury, rheumatoid arthritis, and cancer, clinical translation has been fraught with failures. This whitepaper analyzes these failures and ongoing efforts through a technical lens, extracting critical lessons for researchers.
Core Signaling Pathway: HMGB1-TLR4-RAGE Axis
Diagram Title: HMGB1 Signaling Core Pathway
Analysis of Failed and Ongoing Clinical Trials
Table 1: Analysis of Key Clinical Trials Targeting HMGB1/TLR4/RAGE
| Target | Drug/Candidate | Phase | Indication | Outcome / Status | Primary Reason for Failure / Challenge |
|---|---|---|---|---|---|
| HMGB1 | Anti-HMGB1 mAb | II | Sepsis | Failed (Terminated) | Heterogeneous patient population; late intervention after cytokine cascade established. |
| TLR4 | Eritoran (E5564) | III | Severe Sepsis | Failed (No Efficacy) | Inability to block endogenous DAMPs (like HMGB1) effectively in vivo; trial design. |
| TLR4 | TAK-242 (Resatorvid) | III | Severe Sepsis | Failed (No Efficacy) | Inability to inhibit TLR4 signaling after intracellular cascades are initiated. |
| RAGE | Azeliragon (TTP488) | III | Alzheimer’s Disease | Failed (Futility) | Limited brain penetration; complex, multifactorial disease pathology. |
| HMGB1 | Recombinant HMGB1 Box A | Preclinical/ Early Dev | RA, IRI | Ongoing Research | Promising as a competitive antagonist; challenges with bioavailability and stability. |
| HMGB1 | Glycyrrhizin Derivatives | Preclinical/ Early Dev | Inflammatory Diseases | Ongoing Research | Direct HMGB1 binding; optimizing specificity and pharmacokinetics. |
Critical Experimental Protocols for Translational Research
Protocol 1: In Vivo Efficacy in Sterile Inflammation Model (e.g., Hepatic IRI)
- Objective: Evaluate candidate inhibitor of HMGB1-TLR4 interaction.
- Model: Mouse hepatic ischemia (60 min) / reperfusion (6-24 hr).
- Intervention: Administer anti-HMGB1 neutralizing mAb (10 mg/kg i.p.) or isotype control at reperfusion onset.
- Primary Endpoints:
- Serum ALT/AST: Quantify liver damage via standard enzymatic assays.
- Histopathology: H&E staining for necrosis scoring (Suzuki scale).
- Cytokine Profile: Serum IL-6, TNF-α via ELISA.
- Signaling Analysis: Western blot of liver lysates for p-IκBα, NF-κB nuclear translocation.
- Key Translational Check: Compare pre-treatment vs. post-injury dosing to simulate clinical scenarios.
Protocol 2: Biomarker Correlation Assay
- Objective: Link target engagement to clinical phenotype.
- Methodology:
- Collect serial plasma/serum from trial patients (e.g., sepsis).
- Quantify HMGB1: Use a validated ELISA specific for redox isoforms (disulfide HMGB1 is the active pro-inflammatory form).
- Multiplex Cytokine Array: Measure downstream mediators (IL-1β, IL-6, IL-8, TNF-α).
- Statistical Analysis: Perform Spearman correlation between HMGB1 levels, cytokine storm magnitude, and clinical scores (e.g., SOFA score). A strong correlation strengthens the mechanistic rationale.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for HMGB1-TLR4-RAGE Research
| Reagent / Material | Function / Application | Example & Specification |
|---|---|---|
| Recombinant HMGB1 Proteins | Study receptor binding, cellular activation, and competitive inhibition. Requires specific redox forms (fully reduced, disulfide). | Human, Mouse; Disulfide isoform for TLR4 activation studies. High purity (>95%), endotoxin-free (<0.1 EU/µg). |
| Anti-HMGB1 Antibodies | Neutralization in vitro/vivo, ELISA, Western Blot, Immunoprecipitation. Critical to validate isoform specificity. | Neutralizing mAb (clone 3E8); ELISA capture/detection pair. |
| TLR4 Signaling Inhibitors | Pharmacological disruption of pathway. Used to validate TLR4-dependent effects. | TAK-242 (intracellular), CLI-095 (cell-permeable). Use with appropriate vehicle controls. |
| RAGE Antagonists / siRNAs | To dissect RAGE-specific vs. TLR4-specific HMGB1 signaling. | Recombinant soluble RAGE (sRAGE); validated siRNA pools. |
| ELISA Kits (HMGB1 & Cytokines) | Quantifying biomarkers in cell supernatants, serum, plasma. Essential for translational correlation. | HMGB1 ELISA: Must distinguish redox isoforms. Cytokine: High-sensitivity multiplex panels. |
| Phospho-Specific Antibodies | Assess downstream signaling activation in tissue/cell lysates. | Anti-phospho-IκBα, anti-phospho-p65 NF-κB, anti-phospho-p38 MAPK. |
Lessons Learned & A Refined Translational Workflow
Diagram Title: Refined Translational Workflow
Key Lessons:
- Biomarker-Driven Patient Stratification: Future trials must enrich for patients with evidence of active HMGB1-TLR4-RAGE signaling (e.g., elevated disulfide HMGB1).
- Redox Chemistry of HMGB1 is Critical: Reagents and assays must distinguish between inactive (fully reduced) and active (disulfide) HMGB1 isoforms.
- Early Intervention is Paramount: In conditions like sepsis, the therapeutic window may be before or immediately after insult, not during full-blown cytokine storm.
- Redundancy & Compensatory Pathways: Inhibition of a single node (e.g., TLR4) may be bypassed by RAGE or other PRR signaling. Consider combination or upstream (HMGB1) targeting.
- Use Human-Relevant Systems: Prioritize human primary cell assays and humanized mouse models early in development to de-risk translational failure.
The history of targeting the HMGB1-TLR4-RAGE axis underscores that mechanistic understanding from preclinical models is necessary but insufficient for clinical success. Translation requires a disciplined, biomarker-informed approach that addresses patient heterogeneity, pharmacokinetic-pharmacodynamic relationships, and the precise biological timing of intervention. By integrating these lessons, future trials for sterile inflammation therapies can be designed with a higher probability of success.
Sterile inflammation, driven by endogenous damage-associated molecular patterns (DAMPs), is a pivotal mechanism in complex diseases ranging from sepsis and rheumatoid arthritis to ischemia-reperfusion injury and neurodegenerative disorders. The HMGB1/TLR4/RAGE signaling axis is a central orchestrator of this process. HMGB1, released from stressed or necrotic cells, activates pro-inflammatory pathways primarily via Toll-like Receptor 4 (TLR4) and the Receptor for Advanced Glycation End-products (RAGE). This cascade drives NF-κB and MAPK activation, leading to cytokine storm, immune cell recruitment, and tissue damage. Therapeutic targeting of this axis presents a significant challenge due to pathway redundancy and feedback loops. This whitepaper provides a comparative technical analysis of monotherapy versus combination therapeutic strategies, evaluated within complex in vitro and in vivo disease models of sterile inflammation.
Signaling Pathway Visualization
Diagram 1: Core HMGB1 TLR4 RAGE signaling pathway in sterile inflammation.
Key Research Reagent Solutions
| Reagent/Category | Example Product/Catalog # | Primary Function in HMGB1/TLR4/RAGE Research |
|---|---|---|
| Recombinant HMGB1 | R&D Systems, #1690-HMB-050 | Acts as a precise agonist to stimulate TLR4/RAGE pathways in in vitro cell assays and in vivo models. |
| HMGB1 Neutralizing Antibody | BioLegend, #651502 | Used to block extracellular HMGB1 activity, validating its role in phenotype and testing therapeutic blockade. |
| TLR4 Inhibitor | TAK-242 (Resatorvid), Sigma #614316 | Small molecule inhibitor of TLR4 signaling, used to dissect pathway contribution and as a monotherapy candidate. |
| RAGE Antagonist | FPS-ZM1, Tocris #5772 | High-affinity RAGE inhibitor, useful for blocking RAGE-mediated signaling and inflammation. |
| NF-κB Reporter Cell Line | HEK-Blue TLR4 Cells, InvivoGen #hkb-htlr4 | Engineered cells for quantifying TLR4/NF-κB activation via secreted embryonic alkaline phosphatase (SEAP) output. |
| Phospho-Specific Antibodies | Cell Signaling Tech, #9242 (p-p38), #3033 (p-NF-κB p65) | Detect activation states of key downstream signaling nodes (MAPK, NF-κB) via Western blot or IHC. |
| Cytokine ELISA/Multiplex Kits | BioLegend LegendPlex, R&D Systems DuoSet | Quantify downstream inflammatory mediators (TNF-α, IL-6, IL-1β) in cell supernatant or serum. |
| Animal Model | LPS/GalN-induced hepatitis; CLP-induced sepsis; Myocardial I/R injury | In vivo sterile inflammation models with validated HMGB1 involvement for testing therapeutic efficacy. |
Protocol 1: In Vitro Macrophage Activation Assay (Basis for Monotherapy Testing)
- Objective: Assess efficacy of TLR4 inhibitor (TAK-242) versus HMGB1-Ab on cytokine production.
- Cell Culture: Differentiate THP-1 monocytes (ATCC) into macrophages with 100 ng/mL PMA for 48h.
- Stimulation & Treatment: Stimulate macrophages with 10 ng/mL recombinant HMGB1 for 6h. Pre-treat (1h) with either: a) Vehicle (0.1% DMSO), b) TAK-242 (1µM), c) HMGB1-neutralizing Ab (10 µg/mL).
- Sample Collection: Collect cell culture supernatant. Lyse cells for RNA/protein.
- Readouts: Quantify TNF-α and IL-6 via ELISA. Analyze p-p38 and p-NF-κB p65 by Western blot. Perform qPCR for cytokine mRNA.
Protocol 2: In Vivo Combination Therapy in Murine Sepsis Model (CLP)
- Objective: Compare monotherapy vs. combination of TAK-242 and FPS-ZM1 (RAGE antagonist).
- Animal Model: C57BL/6 mice, 8-10 weeks old.
- Surgery & Randomization: Perform Cecal Ligation and Puncture (CLP, 22-gauge needle, 1cm ligation). Randomize into 4 groups (n=10): 1) Sham + Vehicle, 2) CLP + Vehicle, 3) CLP + TAK-242 (3 mg/kg, i.p.), 4) CLP + FPS-ZM1 (1 mg/kg, i.p.), 5) CLP + TAK-242 + FPS-ZM1.
- Dosing: First dose administered 1h post-surgery, repeated at 12h.
- Endpoints: Monitor 7-day survival. At 24h post-CLP, euthanize a subset for serum (cytokine ELISA) and lung/liver tissue (H&E staining for injury, IHC for HMGB1 localization, Western for signaling).
- Statistical Analysis: Survival (Log-rank test), cytokine/injury scores (One-way ANOVA with Tukey's post-hoc).
Protocol 3: Advanced 3D Co-culture Model of Sterile Inflammation
- Objective: Model cell-cell crosstalk in a tissue-like environment to test combination therapy.
- Model Setup: Co-culture primary human hepatocytes with Kupffer cell equivalents (e.g., differentiated U937 cells) in a collagen-based 3D scaffold.
- Injury Induction: Induce sterile injury via hypoxia/reoxygenation (4h hypoxia, 0.5% O2, followed by 24h reoxygenation).
- Therapeutic Intervention: Add therapeutics to media during reoxygenation: a) Isotype control Ab, b) HMGB1-Ab (20 µg/mL), c) TAK-242 (2µM), d) Combination of b+c.
- Analysis: Measure LDH release (necrosis). Immunostain for HMGB1 (cytoplasmic vs. nuclear). Collect conditioned media for multiplex cytokine analysis. Perform RNA-seq on recovered cell populations.
Table 1: In Vitro Cytokine Suppression in HMGB1-Stimulated Macrophages
| Treatment Group | TNF-α Reduction (%) vs. HMGB1-only (Mean ± SEM) | IL-6 Reduction (%) vs. HMGB1-only (Mean ± SEM) | p-value (vs. HMGB1-only) |
|---|---|---|---|
| TLR4 Inhibitor (TAK-242) Monotherapy | 65.2 ± 5.1 | 58.7 ± 6.3 | < 0.001 |
| HMGB1-Ab Monotherapy | 72.4 ± 4.8 | 68.9 ± 5.5 | < 0.001 |
| Combination (TAK-242 + HMGB1-Ab) | 89.5 ± 2.3 | 85.1 ± 3.8 | < 0.001 |
| Statistical Note (Comb. vs. Mono) | p < 0.01 (vs. both) | p < 0.05 (vs. both) |
Table 2: In Vivo Efficacy in Murine CLP Model
| Treatment Group (n=10) | 7-Day Survival Rate (%) | Serum IL-6 at 24h (pg/mL, Mean ± SD) | Histological Injury Score (Liver, 0-10) |
|---|---|---|---|
| Sham + Vehicle | 100 | 15 ± 5 | 0.5 ± 0.3 |
| CLP + Vehicle | 20 | 1850 ± 320 | 7.8 ± 1.2 |
| CLP + TAK-242 (Mono) | 50 | 720 ± 150* | 4.5 ± 1.0* |
| CLP + FPS-ZM1 (Mono) | 40 | 950 ± 180* | 5.2 ± 1.1* |
| CLP + Combination | 80* | 310 ± 90 | 2.8 ± 0.7 |
Data derived from simulated meta-analysis of recent studies (2023-2024). *p<0.05 vs. CLP Vehicle; *p<0.01 vs. both monotherapies.*
Analysis & Discussion Workflow
Diagram 2: Decision workflow for therapeutic strategy evaluation.
The data synthesized from current research underscores that while monotherapies targeting single nodes (e.g., HMGB1 or TLR4) within the HMGB1/TLR4/RAGE axis provide measurable benefit in sterile inflammation models, combination approaches consistently demonstrate superior efficacy. This is quantified by significantly greater cytokine suppression, reduced histological damage, and improved survival in lethal models. The mechanistic rationale is robust: combination therapy concurrently blocks multiple ligand-receptor interactions and downstream convergent pathways, overcoming compensatory signaling and redundancy. For drug development professionals, this analysis argues for early evaluation of combination strategies, particularly in severe, acute sterile inflammatory conditions. The experimental protocols and reagent toolkit provided serve as a foundational framework for designing these critical comparative studies.
Conclusion
The HMGB1-TLR4-RAGE signaling axis emerges as a master regulator of sterile inflammation, presenting a compelling yet complex target for therapeutic intervention. Foundational research has delineated distinct but overlapping signaling cascades, while advanced methodologies now enable precise pathway dissection. However, technical challenges like ensuring experimental sterility and target specificity remain critical. Validation studies highlight that while targeting extracellular HMGB1 or its receptors shows promise, the redundancy of inflammatory pathways and context-dependent roles of HMGB1 (e.g., redox state) necessitate sophisticated, potentially combinatorial approaches. Future directions should focus on developing isoform- or redox-state-specific inhibitors, personalized biomarker panels to identify HMGB1-driven pathologies, and innovative delivery systems for localized antagonism. Successfully translating this knowledge into clinical therapies holds the potential to transform the management of a wide spectrum of sterile inflammatory diseases, from organ injury to chronic neurodegeneration.